Phase transitions of the pulmonary surfactant film at the perfluorocarbon-water interface
- PMID: 37041745
- PMCID: PMC10209028
- DOI: 10.1016/j.bpj.2023.04.010
Phase transitions of the pulmonary surfactant film at the perfluorocarbon-water interface
Abstract
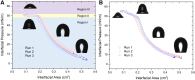
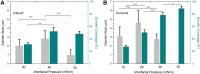
Pulmonary surfactant is a lipid-protein complex that forms a thin film at the air-water surface of the lungs. This surfactant film defines the elastic recoil and respiratory mechanics of the lungs. One generally accepted rationale of using oxygenated perfluorocarbon (PFC) as a respiratory medium in liquid ventilation is to take advantage of its low surface tensions (14-18 mN/m), which was believed to make PFC an ideal replacement of the exogenous surfactant. Compared with the extensive studies of the phospholipid phase behavior of the pulmonary surfactant film at the air-water surface, its phase behavior at the PFC-water interface is essentially unknown. Here, we reported the first detailed biophysical study of phospholipid phase transitions in two animal-derived natural pulmonary surfactant films, Infasurf and Survanta, at the PFC-water interface using constrained drop surfactometry. Constrained drop surfactometry allows in situ Langmuir-Blodgett transfer from the PFC-water interface, thus permitting direct visualization of lipid polymorphism in pulmonary surfactant films using atomic force microscopy. Our data suggested that regardless of its low surface tension, the PFC cannot be used as a replacement of pulmonary surfactant in liquid ventilation where the air-water surface of the lungs is replaced with the PFC-water interface that features an intrinsically high interfacial tension. The pulmonary surfactant film at the PFC-water interface undergoes continuous phase transitions at surface pressures less than the equilibrium spreading pressure of 50 mN/m and a monolayer-to-multilayer transition above this critical pressure. These results provided not only novel biophysical insight into the phase behavior of natural pulmonary surfactant at the oil-water interface but also translational implications into the further development of liquid ventilation and liquid breathing techniques.
Copyright © 2023 Biophysical Society. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests The authors declare no competing interests.
Figures



Similar articles
-
Biophysical function of pulmonary surfactant in liquid ventilation.Biophys J. 2023 Aug 8;122(15):3099-3107. doi: 10.1016/j.bpj.2023.06.014. Epub 2023 Jun 23. Biophys J. 2023. PMID: 37353933 Free PMC article.
-
Atomic Force Microscopy Imaging of Adsorbed Pulmonary Surfactant Films.Biophys J. 2020 Aug 18;119(4):756-766. doi: 10.1016/j.bpj.2020.06.033. Epub 2020 Jul 14. Biophys J. 2020. PMID: 32702292 Free PMC article.
-
Comparative biophysical study of clinical surfactants using constrained drop surfactometry.Am J Physiol Lung Cell Mol Physiol. 2024 Oct 1;327(4):L535-L546. doi: 10.1152/ajplung.00058.2024. Epub 2024 Aug 19. Am J Physiol Lung Cell Mol Physiol. 2024. PMID: 39159363
-
Formation and structure of surface films: captive bubble surfactometry.Biochim Biophys Acta. 1998 Nov 19;1408(2-3):180-202. doi: 10.1016/s0925-4439(98)00067-2. Biochim Biophys Acta. 1998. PMID: 9813315 Review.
-
Protein-lipid interactions and surface activity in the pulmonary surfactant system.Chem Phys Lipids. 2006 Jun;141(1-2):105-18. doi: 10.1016/j.chemphyslip.2006.02.017. Epub 2006 Mar 20. Chem Phys Lipids. 2006. PMID: 16600200 Review.
Cited by
-
Biophysical function of pulmonary surfactant in liquid ventilation.Biophys J. 2023 Aug 8;122(15):3099-3107. doi: 10.1016/j.bpj.2023.06.014. Epub 2023 Jun 23. Biophys J. 2023. PMID: 37353933 Free PMC article.
References
-
- Zuo Y.Y., Veldhuizen R.A.W., et al. Possmayer F. Current perspectives in pulmonary surfactant — inhibition, enhancement and evaluation. Biochim. Biophys. Acta. 2008;1778:1947–1977. - PubMed
-
- Parra E., Pérez-Gil J. Composition, structure and mechanical properties define performance of pulmonary surfactant membranes and films. Chem. Phys. Lipids. 2015;185:153–175. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

