Bioinspired Framework Catalysts: From Enzyme Immobilization to Biomimetic Catalysis
- PMID: 37043332
- PMCID: PMC10853941
- DOI: 10.1021/acs.chemrev.2c00879
Bioinspired Framework Catalysts: From Enzyme Immobilization to Biomimetic Catalysis
Abstract
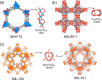
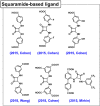
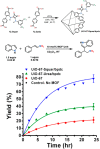
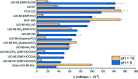
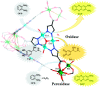
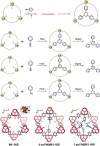
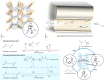
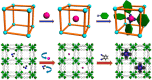
Enzymatic catalysis has fueled considerable interest from chemists due to its high efficiency and selectivity. However, the structural complexity and vulnerability hamper the application potentials of enzymes. Driven by the practical demand for chemical conversion, there is a long-sought quest for bioinspired catalysts reproducing and even surpassing the functions of natural enzymes. As nanoporous materials with high surface areas and crystallinity, metal-organic frameworks (MOFs) represent an exquisite case of how natural enzymes and their active sites are integrated into porous solids, affording bioinspired heterogeneous catalysts with superior stability and customizable structures. In this review, we comprehensively summarize the advances of bioinspired MOFs for catalysis, discuss the design principle of various MOF-based catalysts, such as MOF-enzyme composites and MOFs embedded with active sites, and explore the utility of these catalysts in different reactions. The advantages of MOFs as enzyme mimetics are also highlighted, including confinement, templating effects, and functionality, in comparison with homogeneous supramolecular catalysts. A perspective is provided to discuss potential solutions addressing current challenges in MOF catalysis.
Conflict of interest statement
The authors declare no competing financial interest.
Figures









































Similar articles
-
Site Isolation in Metal-Organic Frameworks Enables Novel Transition Metal Catalysis.Acc Chem Res. 2018 Sep 18;51(9):2129-2138. doi: 10.1021/acs.accounts.8b00297. Epub 2018 Aug 21. Acc Chem Res. 2018. PMID: 30129753
-
[Advances in enzyme immobilization based on hierarchical porous metal-organic frameworks].Sheng Wu Gong Cheng Xue Bao. 2023 Mar 25;39(3):930-941. doi: 10.13345/j.cjb.220623. Sheng Wu Gong Cheng Xue Bao. 2023. PMID: 36994563 Review. Chinese.
-
Porous metal-organic frameworks for heterogeneous biomimetic catalysis.Acc Chem Res. 2014 Apr 15;47(4):1199-207. doi: 10.1021/ar400265x. Epub 2014 Feb 6. Acc Chem Res. 2014. PMID: 24499017
-
Hybrid Metal-Organic Frameworks (MOFs) for Various Catalysis Applications.Top Curr Chem (Cham). 2024 Dec 13;383(1):3. doi: 10.1007/s41061-024-00486-5. Top Curr Chem (Cham). 2024. PMID: 39671137 Review.
-
Enzyme Immobilization on Metal-Organic Framework (MOF): Effects on Thermostability and Function.Protein Pept Lett. 2019;26(9):636-647. doi: 10.2174/0929866526666190430120046. Protein Pept Lett. 2019. PMID: 31208305 Review.
Cited by
-
Selective Hydrolysis of Heterooligosaccharides by Poly(acrylate) Gel Catalysts.ACS Catal. 2024 Oct 30;14(22):16723-16730. doi: 10.1021/acscatal.4c04697. eCollection 2024 Nov 15. ACS Catal. 2024. PMID: 39569156 Free PMC article.
-
Recent Advances in Enzymatic Biofuel Cells to Power Up Wearable and Implantable Biosensors.Biosensors (Basel). 2025 Mar 28;15(4):218. doi: 10.3390/bios15040218. Biosensors (Basel). 2025. PMID: 40277532 Free PMC article. Review.
-
CO-Free Aminocarbonylation of Terminal Alkynes Catalyzed by Synergistic Effect From Metal-Organic Frameworks.Adv Sci (Weinh). 2024 Nov;11(41):e2405308. doi: 10.1002/advs.202405308. Epub 2024 Sep 5. Adv Sci (Weinh). 2024. PMID: 39234812 Free PMC article.
-
Molecularly imprinted MOF nanozymes: Demonstration of smartphone-integrated dual-mode platform for ratiometric fluorescent/colorimetric detection of chloramphenicol.Food Chem X. 2025 Feb 25;26:102322. doi: 10.1016/j.fochx.2025.102322. eCollection 2025 Feb. Food Chem X. 2025. PMID: 40104617 Free PMC article.
-
Safety Landscape of Therapeutic Nanozymes and Future Research Directions.Adv Sci (Weinh). 2024 Dec;11(46):e2407816. doi: 10.1002/advs.202407816. Epub 2024 Oct 24. Adv Sci (Weinh). 2024. PMID: 39445544 Free PMC article. Review.
References
-
- Dawson J. H.; Sono M. Cytochrome P-450 and Chloroperoxidase: Thiolate-Ligated Heme Enzymes. Spectroscopic Determination of Their Active-Site Structures and Mechanistic Implications of Thiolate Ligation. Chem. Rev. 1987, 87, 1255–1276. 10.1021/cr00081a015. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

