Lysophosphatidic acid stimulates rat uterine contraction in vitro
- PMID: 37045747
- PMCID: PMC10267583
- DOI: 10.1262/jrd.2023-011
Lysophosphatidic acid stimulates rat uterine contraction in vitro
Abstract
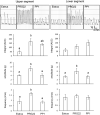
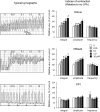
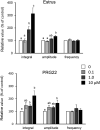
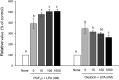
Lysophosphatidic acid (LPA) has been implicated in the uterine endometrial functions of implantation and decidualization; however, not much is known about its myometrial contractile function. Herein we characterized the uterotonic effects of LPA in non-pregnant (estrus) and peri-parturient rats in vitro. LPA dose-dependently (0.01-10 μM) stimulated the amplitude and integral, but not the frequency, of the uterine strip contraction of estrous rats. The stimulatory effect of LPA was enhanced 1 day before parturition but was lost 1 day postpartum. LPA did not cause the de novo synthesis of prostaglandin (PG) F2α but stimulated contractions cooperatively with the PG. LPA-induced contractions were significantly inhibited by an LPA1/2/3 antagonist in the uteri of estrous rats but not in term rats. This study characterized the uterotonic effect of a natural LPA that occurs at physiological concentrations, changes with reproductive states, and is independent of mediation by the newly synthesized PG.
Keywords: Contraction; Lysophosphatidic acid; Prostaglandin F2α; Uterus.
Conflict of interest statement
The authors have nothing to disclose.
Figures
Similar articles
-
TXA2 mediates LPA1-stimulated uterine contraction in late pregnant mouse.Prostaglandins Other Lipid Mediat. 2023 Aug;167:106736. doi: 10.1016/j.prostaglandins.2023.106736. Epub 2023 Apr 14. Prostaglandins Other Lipid Mediat. 2023. PMID: 37062326
-
Participation of analogues of lysophosphatidic acid (LPA): oleoyl-sn-glycero-3-phosphate (L-alpha-LPA) and 1-oleoyl-2-O-methyl-rac-glycerophosphothionate (OMPT) in uterine smooth muscle contractility of the pregnant pigs.Pol J Vet Sci. 2012;15(4):635-43. doi: 10.2478/v10181-012-0100-9. Pol J Vet Sci. 2012. PMID: 23390752
-
Analysis of lysophosphatidic acid (LPA) receptor and LPA-induced endometrial prostaglandin-endoperoxide synthase 2 expression in the porcine uterus.Endocrinology. 2008 Dec;149(12):6166-75. doi: 10.1210/en.2008-0354. Epub 2008 Aug 14. Endocrinology. 2008. PMID: 18703629
-
Uterine blood supply as a main factor involved in the regulation of the estrous cycle--a new theory.Reprod Biol. 2002 Jul;2(2):93-114. Reprod Biol. 2002. PMID: 14666152 Review.
-
Lysophosphatidic acid (LPA) signaling in human and ruminant reproductive tract.Mediators Inflamm. 2014;2014:649702. doi: 10.1155/2014/649702. Epub 2014 Mar 12. Mediators Inflamm. 2014. PMID: 24744506 Free PMC article. Review.
Cited by
-
The effect of lysophosphatidic acid on myometrial contractility and the mRNA transcription of its receptors in the myometrium at different stages of endometrosis in mares.BMC Vet Res. 2024 Dec 19;20(1):571. doi: 10.1186/s12917-024-04384-2. BMC Vet Res. 2024. PMID: 39696406 Free PMC article.
-
Evaluation of Lysophosphatidic Acid Effects and Its Receptors During Bovine Embryo Development.Int J Mol Sci. 2025 Mar 13;26(6):2596. doi: 10.3390/ijms26062596. Int J Mol Sci. 2025. PMID: 40141238 Free PMC article.
-
Expression of lysophosphatidic acid receptors in the rat uterus: cellular distribution of protein and gestation-associated changes in gene expression.J Vet Med Sci. 2023 Nov 2;85(11):1165-1171. doi: 10.1292/jvms.23-0336. Epub 2023 Oct 2. J Vet Med Sci. 2023. PMID: 37779089 Free PMC article.
-
Imaging the dynamics of murine uterine contractions in early pregnancy†.Biol Reprod. 2024 Jun 12;110(6):1175-1190. doi: 10.1093/biolre/ioae071. Biol Reprod. 2024. PMID: 38713674 Free PMC article.
-
Autotaxin Expression in the Uterus of Cycling Rats.Dev Reprod. 2024 Sep;28(3):67-74. doi: 10.12717/DR.2024.28.3.67. Epub 2024 Sep 30. Dev Reprod. 2024. PMID: 39444640 Free PMC article.
References
-
- Aikawa S, Hashimoto T, Kano K, Aoki J. Lysophosphatidic acid as a lipid mediator with multiple biological actions. J Biochem 2015; 157: 81–89. - PubMed
-
- Nagamatsu T, Iwasawa-Kawai Y, Ichikawa M, Kawana K, Yamashita T, Osuga Y, Fujii T, Schust DJ. Emerging roles for lysophospholipid mediators in pregnancy. Am J Reprod Immunol 2014; 72: 182–191. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

