Bowel Preparation for Colonoscopy Changes Serum Composition as Detected by Thermal Liquid Biopsy and Fluorescence Spectroscopy
- PMID: 37046613
- PMCID: PMC10093451
- DOI: 10.3390/cancers15071952
Bowel Preparation for Colonoscopy Changes Serum Composition as Detected by Thermal Liquid Biopsy and Fluorescence Spectroscopy
Abstract
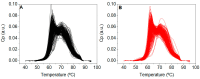
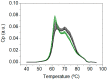
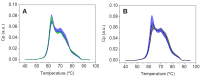
(1) Background: About 50% of prescribed colonoscopies report no pathological findings. A secondary screening test after fecal immunochemical test positivity (FIT+) would be required. Considering thermal liquid biopsy (TLB) as a potential secondary test, the aim of this work was to study possible interferences of colonoscopy bowel preparation on TLB outcome on a retrospective study; (2) Methods: Three groups were studied: 1/514 FIT(+) patients enrolled in a colorectal screening program (CN and CP with normal and pathological colonoscopy, respectively), with blood samples obtained just before colonoscopy and after bowel preparation; 2/55 patients from the CN group with blood sample redrawn after only standard 8-10 h fasting and no bowel preparation (CNR); and 3/55 blood donors from the biobank considered as a healthy control group; (3) Results: The results showed that from the 514 patients undergoing colonoscopy, 247 had CN and 267 had CP. TLB parameters in these two groups were similar but different from those of the blood donors. The resampled patients (with normal colonoscopy and no bowel preparation) had similar TLB parameters to those of the blood donors. TLB parameters together with fluorescence spectra and other serum indicators (albumin and C-reactive protein) confirmed the statistically significant differences between normal colonoscopy patients with and without bowel preparation; (4) Conclusions: Bowel preparation seemed to alter serum protein levels and altered TLB parameters (different from a healthy subject). The diagnostic capability of other liquid-biopsy-based methods might also be compromised. Blood extraction after bowel preparation for colonoscopy should be avoided.
Keywords: colonoscopy; colorectal cancer; fluorescence spectroscopy; thermal liquid biopsy.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Fecal occult blood test for colorectal cancer screening: an evidence-based analysis.Ont Health Technol Assess Ser. 2009;9(10):1-40. Epub 2009 Sep 1. Ont Health Technol Assess Ser. 2009. PMID: 23074514 Free PMC article.
-
Nurse-led reinforced education by mobile messenger improves the quality of bowel preparation of colonoscopy in a population-based colorectal cancer screening program: A randomized controlled trial.Int J Nurs Stud. 2022 Sep;133:104301. doi: 10.1016/j.ijnurstu.2022.104301. Epub 2022 May 30. Int J Nurs Stud. 2022. PMID: 35764027 Clinical Trial.
-
Diagnostic yield of early repeat colonoscopy after suboptimal bowel preparation in a fecal immunochemical test-based screening program.Endoscopy. 2020 Dec;52(12):1093-1100. doi: 10.1055/a-1191-3011. Epub 2020 Jun 24. Endoscopy. 2020. PMID: 32583393
-
Efficacy and tolerability of a low-residue diet for bowel preparation: systematic review and meta-analysis.Surg Endosc. 2022 Jun;36(6):3858-3875. doi: 10.1007/s00464-021-08703-8. Epub 2021 Sep 1. Surg Endosc. 2022. PMID: 34471982
-
Bowel Preparation for Colonoscopy in 2020: A Look at the Past, Present, and Future.Curr Gastroenterol Rep. 2020 May 6;22(6):28. doi: 10.1007/s11894-020-00764-4. Curr Gastroenterol Rep. 2020. PMID: 32377915 Review.
Cited by
-
The development of an ingestible biosensor for the characterization of gut metabolites related to major depressive disorder: hypothesis and theory.Front Syst Biol. 2023 Dec 5;3:1274184. doi: 10.3389/fsysb.2023.1274184. eCollection 2023. Front Syst Biol. 2023. PMID: 40809492 Free PMC article.
References
-
- Gini A., Jansen E.E.L., Zielonke N., Meester R.G.S., Senore C., Anttila A., Segnan N., Mlakar D.N., de Koning H.J., Lansdorp-Vogelaar I., et al. Impact of Colorectal Cancer Screening on Cancer-Specific Mortality in Europe: A Systematic Review. Eur. J. Cancer. 2020;127:224–235. doi: 10.1016/j.ejca.2019.12.014. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

