Novel Cannabinoid Receptor 2 (CB2) Low Lipophilicity Agonists Produce Distinct cAMP and Arrestin Signalling Kinetics without Bias
- PMID: 37047385
- PMCID: PMC10094510
- DOI: 10.3390/ijms24076406
Novel Cannabinoid Receptor 2 (CB2) Low Lipophilicity Agonists Produce Distinct cAMP and Arrestin Signalling Kinetics without Bias
Abstract
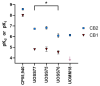
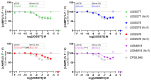
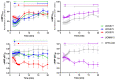
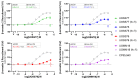
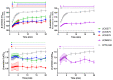
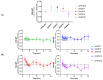
Cannabinoid Receptor 2 (CB2) is a promising target for treating inflammatory diseases. We designed derivatives of 3-carbamoyl-2-pyridone and 1,8-naphthyridin-2(1H)-one-3-carboxamide CB2-selective agonists with reduced lipophilicity. The new compounds were measured for their affinity (radioligand binding) and ability to elicit cyclic adenosine monophosphate (cAMP) signalling and β-arrestin-2 translocation with temporal resolution (BRET-based biosensors). For the 3-carbamoyl-2-pyridone derivatives, we found that modifying the previously reported compound UOSS77 (also known as S-777469) by appending a PEG2-alcohol via a 3-carbomylcyclohexyl carboxamide (UOSS75) lowered lipophilicity, and preserved binding affinity and signalling profile. The 1,8-naphthyridin-2(1H)-one-3-carboxamide UOMM18, containing a cis configuration at the 3-carboxamide cyclohexyl and with an alcohol on the 4-position of the cyclohexyl, had lower lipophilicity but similar CB2 affinity and biological activity to previously reported compounds of this class. Relative to CP55,940, the new compounds acted as partial agonists and did not exhibit signalling bias. Interestingly, while all compounds shared similar temporal trajectories for maximal efficacy, differing temporal trajectories for potency were observed. Consequently, when applied at sub-maximal concentrations, CP55,940 tended to elicit sustained (cAMP) or increasing (arrestin) responses, whereas responses to the new compounds tended to be transient (cAMP) or sustained (arrestin). In future studies, the compounds characterised here may be useful in elucidating the consequences of differential temporal signalling profiles on CB2-mediated physiological responses.
Keywords: CB2; beta-arrestin 2; cannabinoid; cannabinoid receptor agonists; chemistry; cyclic AMP; drug development; kinetics; pharmaceutical; receptor; signal transduction.
Conflict of interest statement
The authors declare they have no conflict of interest to disclose.
Figures
Similar articles
-
Signalling profiles of a structurally diverse panel of synthetic cannabinoid receptor agonists.Biochem Pharmacol. 2020 May;175:113871. doi: 10.1016/j.bcp.2020.113871. Epub 2020 Feb 21. Biochem Pharmacol. 2020. PMID: 32088263
-
Investigating selectivity and bias for G protein subtypes and β-arrestins by synthetic cannabinoid receptor agonists at the cannabinoid CB1 receptor.Biochem Pharmacol. 2024 Apr;222:116052. doi: 10.1016/j.bcp.2024.116052. Epub 2024 Feb 12. Biochem Pharmacol. 2024. PMID: 38354957
-
Design, synthesis, and structure-activity relationships of diindolylmethane derivatives as cannabinoid CB2 receptor agonists.Arch Pharm (Weinheim). 2023 Mar;356(3):e2200493. doi: 10.1002/ardp.202200493. Epub 2022 Nov 27. Arch Pharm (Weinheim). 2023. PMID: 36437108
-
Cannabinoid CB1 and CB2 receptor ligand specificity and the development of CB2-selective agonists.Curr Med Chem. 2008;15(14):1428-43. doi: 10.2174/092986708784567716. Curr Med Chem. 2008. PMID: 18537620 Review.
-
Functional selectivity at G-protein coupled receptors: Advancing cannabinoid receptors as drug targets.Biochem Pharmacol. 2017 Mar 15;128:1-11. doi: 10.1016/j.bcp.2016.11.014. Epub 2016 Nov 24. Biochem Pharmacol. 2017. PMID: 27890725 Free PMC article. Review.
Cited by
-
Challenges and Future Trends in Atopic Dermatitis.Int J Mol Sci. 2023 Jul 12;24(14):11380. doi: 10.3390/ijms241411380. Int J Mol Sci. 2023. PMID: 37511138 Free PMC article. Review.
References
-
- Manera C., Saccomanni G., Adinolfi B., Benetti V., Ligresti A., Cascio M.G., Tuccinardi T., Lucchesi V., Martinelli A., Nieri P., et al. Rational Design, Synthesis, and Pharmacological Properties of New 1,8-Naphthyridin-2(1H)-on-3-Carboxamide Derivatives as Highly Selective Cannabinoid-2 Receptor Agonists. J. Med. Chem. 2009;52:3644–3651. doi: 10.1021/jm801563d. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

