Molecular and Functional Characteristics of Airway Epithelium under Chronic Hypoxia
- PMID: 37047450
- PMCID: PMC10095024
- DOI: 10.3390/ijms24076475
Molecular and Functional Characteristics of Airway Epithelium under Chronic Hypoxia
Abstract
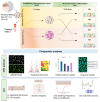
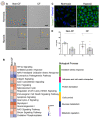
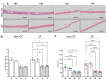
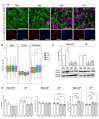
Localized and chronic hypoxia of airway mucosa is a common feature of progressive respiratory diseases, including cystic fibrosis (CF). However, the impact of prolonged hypoxia on airway stem cell function and differentiated epithelium is not well elucidated. Acute hypoxia alters the transcription and translation of many genes, including the CF transmembrane conductance regulator (CFTR). CFTR-targeted therapies (modulators) have not been investigated in vitro under chronic hypoxic conditions found in CF airways in vivo. Nasal epithelial cells (hNECs) derived from eight CF and three non-CF participants were expanded and differentiated at the air-liquid interface (26-30 days) at ambient and 2% oxygen tension (hypoxia). Morphology, global proteomics (LC-MS/MS) and function (barrier integrity, cilia motility and ion transport) of basal stem cells and differentiated cultures were assessed. hNECs expanded at chronic hypoxia, demonstrating epithelial cobblestone morphology and a similar proliferation rate to hNECs expanded at normoxia. Hypoxia-inducible proteins and pathways in stem cells and differentiated cultures were identified. Despite the stem cells' plasticity and adaptation to chronic hypoxia, the differentiated epithelium was significantly thinner with reduced barrier integrity. Stem cell lineage commitment shifted to a more secretory epithelial phenotype. Motile cilia abundance, length, beat frequency and coordination were significantly negatively modulated. Chronic hypoxia reduces the activity of epithelial sodium and CFTR ion channels. CFTR modulator drug response was diminished. Our findings shed light on the molecular pathophysiology of hypoxia and its implications in CF. Targeting hypoxia can be a strategy to augment mucosal function and may provide a means to enhance the efficacy of CFTR modulators.
Keywords: CFTR; airway stem cell; cystic fibrosis; hypoxia.
Conflict of interest statement
SAW is the recipient of a Vertex Innovation Grant (2018) and a TSANZ/Vertex Research Award (2020). Both are unrelated and outside of the submitted manuscript. AJ has received consulting fees from Vertex on projects unrelated to this study. All other authors declare no conflict of interest.
Figures


Similar articles
-
Significant functional differences in differentiated Conditionally Reprogrammed (CRC)- and Feeder-free Dual SMAD inhibited-expanded human nasal epithelial cells.J Cyst Fibros. 2021 Mar;20(2):364-371. doi: 10.1016/j.jcf.2020.12.019. Epub 2021 Jan 5. J Cyst Fibros. 2021. PMID: 33414087
-
Characterization of pediatric cystic fibrosis airway epithelial cell cultures at the air-liquid interface obtained by non-invasive nasal cytology brush sampling.Respir Res. 2017 Dec 28;18(1):215. doi: 10.1186/s12931-017-0706-7. Respir Res. 2017. PMID: 29282053 Free PMC article.
-
CFTR delivery to 25% of surface epithelial cells restores normal rates of mucus transport to human cystic fibrosis airway epithelium.PLoS Biol. 2009 Jul;7(7):e1000155. doi: 10.1371/journal.pbio.1000155. Epub 2009 Jul 21. PLoS Biol. 2009. PMID: 19621064 Free PMC article.
-
CFTR Modulator Therapies: Potential Impact on Airway Infections in Cystic Fibrosis.Cells. 2022 Apr 6;11(7):1243. doi: 10.3390/cells11071243. Cells. 2022. PMID: 35406809 Free PMC article. Review.
-
Cystic Fibrosis Transmembrane Conductance Regulator Modulator Therapy: A Review for the Otolaryngologist.Am J Rhinol Allergy. 2020 Jul;34(4):573-580. doi: 10.1177/1945892420912368. Epub 2020 Mar 13. Am J Rhinol Allergy. 2020. PMID: 32168995 Free PMC article. Review.
Cited by
-
The Impact of NO2 on Epithelial Barrier Integrity of a Primary Cell-Based Air-Liquid Interface Model of the Nasal Respiratory Epithelium.J Appl Toxicol. 2025 Mar;45(3):482-491. doi: 10.1002/jat.4717. Epub 2024 Nov 12. J Appl Toxicol. 2025. PMID: 39529574 Free PMC article.
-
Transcriptomic Analysis of Air-Liquid Interface Culture in Human Lung Organoids Reveals Regulators of Epithelial Differentiation.Cells. 2024 Dec 2;13(23):1991. doi: 10.3390/cells13231991. Cells. 2024. PMID: 39682739 Free PMC article.
References
-
- Enocson A., Jordan R., Adab P., Dickens A., Fitzmaurice D. Prevalence and characteristics of low oxygen saturation (SpO2) in a primary care COPD cohort. Eur. Respir. J. 2016;48:PA3937. doi: 10.1183/13993003.congress-2016.PA3937. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

