Effects of Follicle-Stimulating Hormone on Human Sperm Motility In Vitro
- PMID: 37047508
- PMCID: PMC10095528
- DOI: 10.3390/ijms24076536
Effects of Follicle-Stimulating Hormone on Human Sperm Motility In Vitro
Abstract
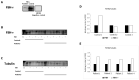
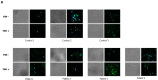
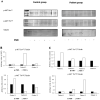
To evaluate whether the follicle-stimulating hormone (FSH) receptor (FSHR) is expressed in human spermatozoa and the effects of FSH incubation on sperm function. Twenty-four Caucasian men were recruited. Thirteen patients had asthenozoospermia, and the remaining 11 had normal sperm parameters (controls). After confirming FSHR expression, spermatozoa from patients and controls were incubated with increasing concentrations of human purified FSH (hpFSH) to reassess FSHR expression and localization and to evaluate progressive and total sperm motility, the mitochondrial membrane potential, and protein kinase B (AKT) 473 and 308 phosphorylation. FSHR is expressed in the post-acrosomal segment, neck, midpiece, and tail of human spermatozoa. Its localization does not differ between patients and controls. Incubation with hpFSH at a concentration of 30 mIU/mL appeared to increase FSHR expression mainly in patients. Incubation of human spermatozoa with hpFSH overall resulted in an overall deterioration of both progressive and total motility in patients and controls and worse mitochondrial function only in controls. Finally, incubation with FSH increased AKT473/tubulin phosphorylation to a greater extent than AKT308. FSHR is expressed in the post-acrosomal region, neck, midpiece, and tail of human spermatozoa. Contrary to a previous study, we report a negative effect of FSH on sperm motility and mitochondrial function. FSH also activates the AKT473 signaling pathway.
Keywords: FSH; FSHR; sperm motility; spermatozoa.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Davis D., Liu X., Segaloff D.L. Identification of the sites of N-linked glycosylation on the follicle-stimulating hormone (FSH) receptor and assessment of their role in FSH receptor function. Mol. Endocrinol. 1995;9:159–170. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

