IVM Advances for Early Antral Follicle-Enclosed Oocytes Coupling Reproductive Tissue Engineering to Inductive Influences of Human Chorionic Gonadotropin and Ovarian Surface Epithelium Coculture
- PMID: 37047595
- PMCID: PMC10095509
- DOI: 10.3390/ijms24076626
IVM Advances for Early Antral Follicle-Enclosed Oocytes Coupling Reproductive Tissue Engineering to Inductive Influences of Human Chorionic Gonadotropin and Ovarian Surface Epithelium Coculture
Abstract
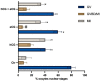
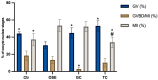
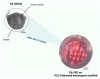
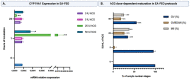
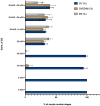
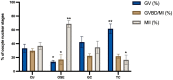
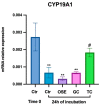
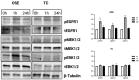
In vitro maturation (IVM) is not a routine assisted reproductive technology (ART) for oocytes collected from early antral (EA) follicles, a large source of potentially available gametes. Despite substantial improvements in IVM in the past decade, the outcomes remain low for EA-derived oocytes due to their reduced developmental competences. To optimize IVM for ovine EA-derived oocytes, a three-dimensional (3D) scaffold-mediated follicle-enclosed oocytes (FEO) system was compared with a validated cumulus-oocyte complex (COC) protocol. Gonadotropin stimulation (eCG and/or hCG) and/or somatic cell coculture (ovarian vs. extraovarian-cell source) were supplied to both systems. The maturation rate and parthenogenetic activation were significantly improved by combining hCG stimulation with ovarian surface epithelium (OSE) cells coculture exclusively on the FEO system. Based on the data, the paracrine factors released specifically from OSE enhanced the hCG-triggering of oocyte maturation mechanisms by acting through the mural compartment (positive effect on FEO and not on COC) by stimulating the EGFR signaling. Overall, the FEO system performed on a developed reproductive scaffold proved feasible and reliable in promoting a synergic cytoplasmatic and nuclear maturation, offering a novel cultural strategy to widen the availability of mature gametes for ART.
Keywords: EGF signaling; PCL electrospun scaffold; early antral follicle; follicle-enclosed oocyte; human chorionic gonadotropin; in vitro oocyte maturation; ovarian surface epithelium; sheep.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Vuong L.N., Ho T.M., Gilchrist R.B., Smitz J. The Place of In Vitro Maturation in Assisted Reproductive Technology. Fertil. Reprod. 2019;1:11–15. doi: 10.1142/S2661318219300022. - DOI
-
- Walls M.L., Hunter T., Ryan J.P., Keelan J.A., Nathan E., Hart R.J. In vitro maturation as an alternative to standard in vitro fertilization for patients diagnosed with polycystic ovaries: A comparative analysis of fresh, frozen and cumulative cycle outcomes. Hum. Reprod. 2015;30:88–96. doi: 10.1093/humrep/deu248. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

