Different Alterations of Hippocampal and Reticulo-Thalamic GABAergic Parvalbumin-Expressing Interneurons Underlie Different States of Unconsciousness
- PMID: 37047741
- PMCID: PMC10094978
- DOI: 10.3390/ijms24076769
Different Alterations of Hippocampal and Reticulo-Thalamic GABAergic Parvalbumin-Expressing Interneurons Underlie Different States of Unconsciousness
Abstract
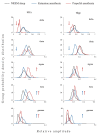
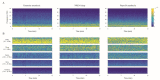
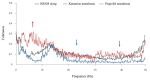
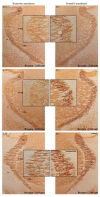
We traced the changes in GABAergic parvalbumin (PV)-expressing interneurons of the hippocampus and reticulo-thalamic nucleus (RT) as possible underlying mechanisms of the different local cortical and hippocampal electroencephalographic (EEG) microstructures during the non-rapid-eye movement (NREM) sleep compared with anesthesia-induced unconsciousness by two anesthetics with different main mechanisms of action (ketamine/diazepam versus propofol). After 3 h of recording their sleep, the rats were divided into two experimental groups: one half received ketamine/diazepam anesthesia and the other half received propofol anesthesia. We simultaneously recorded the EEG of the motor cortex and hippocampus during sleep and during 1 h of surgical anesthesia. We performed immunohistochemistry and analyzed the PV and postsynaptic density protein 95 (PSD-95) expression. PV suppression in the hippocampus and at RT underlies the global theta amplitude attenuation and hippocampal gamma augmentation that is a unique feature of ketamine-induced versus propofol-induced unconsciousness and NREM sleep. While PV suppression resulted in an increase in hippocampal PSD-95 expression, there was no imbalance between inhibition and excitation during ketamine/diazepam anesthesia compared with propofol anesthesia in RT. This increased excitation could be a consequence of a lower GABA interneuronal activity and an additional mechanism underlying the unique local EEG microstructure in the hippocampus during ketamine/diazepam anesthesia.
Keywords: EEG microstructure; GABAergic parvalbumin-expressing interneurons; anesthesia; hippocampus; postsynaptic density protein 95 (PSD-95); reticulo-thalamic nucleus; sleep; unconsciousness.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
-
- Vanini G., Bassana M., Mast M., Mondino A., Cerda I., Phyle M., Chen V., Colmenero A.V., Hambrecht-Wiedbusch V.S., Mashour G.A. Activation of preoptic GABAergic or glutamatergic neurons modulates sleep-wake architecture but not anesthetic state transitions. Curr. Biol. 2020;30:779–787. doi: 10.1016/j.cub.2019.12.063. - DOI - PMC - PubMed
-
- Date A., Bashir K., Uddin A., Nigam C. Differences between natural sleep and the anesthetic state. Future Sci. OA. 2020;6:FSO664. doi: 10.2144/fsoa-2020-0149. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

