Tau Transfer via Extracellular Vesicles Disturbs the Astrocytic Mitochondrial System
- PMID: 37048058
- PMCID: PMC10093208
- DOI: 10.3390/cells12070985
Tau Transfer via Extracellular Vesicles Disturbs the Astrocytic Mitochondrial System
Abstract
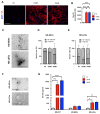
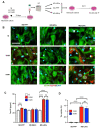
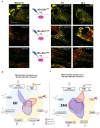
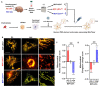
Tauopathies are neurodegenerative disorders involving the accumulation of tau isoforms in cell subpopulations such as astrocytes. The origins of the 3R and 4R isoforms of tau that accumulate in astrocytes remain unclear. Extracellular vesicles (EVs) were isolated from primary neurons overexpressing 1N3R or 1N4R tau or from human brain extracts (progressive supranuclear palsy or Pick disease patients or controls) and characterized (electron microscopy, nanoparticle tracking analysis (NTA), proteomics). After the isolated EVs were added to primary astrocytes or human iPSC-derived astrocytes, tau transfer and mitochondrial system function were evaluated (ELISA, immunofluorescence, MitoTracker staining). We demonstrated that neurons in which 3R or 4R tau accumulated had the capacity to transfer tau to astrocytes and that EVs were essential for the propagation of both isoforms of tau. Treatment with tau-containing EVs disrupted the astrocytic mitochondrial system, altering mitochondrial morphology, dynamics, and redox state. Although similar levels of 3R and 4R tau were transferred, 3R tau-containing EVs were significantly more damaging to astrocytes than 4R tau-containing EVs. Moreover, EVs isolated from the brain fluid of patients with different tauopathies affected mitochondrial function in astrocytes derived from human iPSCs. Our data indicate that tau pathology spreads to surrounding astrocytes via EVs-mediated transfer and modifies their function.
Keywords: astrocytes; extracellular vesicles; mitochondria; tau spreading; tauopathies.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

