Substances of Natural Origin in Medicine: Plants vs. Cancer
- PMID: 37048059
- PMCID: PMC10092955
- DOI: 10.3390/cells12070986
Substances of Natural Origin in Medicine: Plants vs. Cancer
Abstract
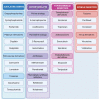
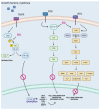
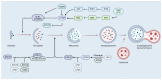
Continuous monitoring of the population's health is the main method of learning about disease prevalence. National and international data draw attention to the persistently high rates of cancer incidence. This necessitates the intensification of efforts aimed at developing new, more effective chemotherapeutic and chemopreventive drugs. Plants represent an invaluable source of natural substances with versatile medicinal properties. Multidirectional activities exhibited by natural substances and their ability to modulate key signaling pathways, mainly related to cancer cell death, make these substances an important research direction. This review summarizes the information regarding plant-derived chemotherapeutic drugs, including their mechanisms of action, with a special focus on selected anti-cancer drugs (paclitaxel, irinotecan) approved in clinical practice. It also presents promising plant-based drug candidates currently being tested in clinical and preclinical trials (betulinic acid, resveratrol, and roburic acid).
Keywords: betulinic acid; cancer; irinotecan; paclitaxel; resveratrol; roburic acid.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Cancer chemopreventive and therapeutic potential of resveratrol: mechanistic perspectives.Cancer Lett. 2008 Oct 8;269(2):243-61. doi: 10.1016/j.canlet.2008.03.057. Epub 2008 Jun 11. Cancer Lett. 2008. PMID: 18550275 Review.
-
Bioactive natural products in cancer prevention and therapy: Progress and promise.Semin Cancer Biol. 2016 Oct;40-41:1-3. doi: 10.1016/j.semcancer.2016.08.006. Epub 2016 Aug 23. Semin Cancer Biol. 2016. PMID: 27565447
-
Cell death mechanisms of plant-derived anticancer drugs: beyond apoptosis.Apoptosis. 2015 Dec;20(12):1531-62. doi: 10.1007/s10495-015-1169-2. Apoptosis. 2015. PMID: 26362468 Review.
-
Chemotherapeutic potential of the chemopreventive phytoalexin resveratrol.Drug Resist Updat. 2004 Dec;7(6):333-44. doi: 10.1016/j.drup.2004.11.001. Epub 2004 Dec 19. Drug Resist Updat. 2004. PMID: 15790544 Review.
-
Resveratrol Action on Lipid Metabolism in Cancer.Int J Mol Sci. 2019 Jun 1;20(11):2704. doi: 10.3390/ijms20112704. Int J Mol Sci. 2019. PMID: 31159437 Free PMC article. Review.
Cited by
-
Exploring the Comprehensive Neuroprotective and Anticancer Potential of Afzelin.Pharmaceuticals (Basel). 2024 May 28;17(6):701. doi: 10.3390/ph17060701. Pharmaceuticals (Basel). 2024. PMID: 38931368 Free PMC article. Review.
-
Biodegradable Persistent Luminescence Nanoparticles as Pyroptosis Inducer for High-Efficiency Tumor Immunotherapy.Adv Sci (Weinh). 2024 Oct;11(39):e2406340. doi: 10.1002/advs.202406340. Epub 2024 Aug 19. Adv Sci (Weinh). 2024. PMID: 39158490 Free PMC article.
-
Anti-Cancer Properties of Resveratrol: A Focus on Its Impact on Mitochondrial Functions.Antioxidants (Basel). 2023 Nov 29;12(12):2056. doi: 10.3390/antiox12122056. Antioxidants (Basel). 2023. PMID: 38136176 Free PMC article. Review.
-
SREBPs as the potential target for solving the polypharmacy dilemma.Front Physiol. 2024 Jan 10;14:1272540. doi: 10.3389/fphys.2023.1272540. eCollection 2023. Front Physiol. 2024. PMID: 38269061 Free PMC article. Review.
-
Caffeic Acid Phenethyl Ester (CAPE) Synergistically Enhances Paclitaxel Activity in Ovarian Cancer Cells.Molecules. 2023 Aug 1;28(15):5813. doi: 10.3390/molecules28155813. Molecules. 2023. PMID: 37570782 Free PMC article.
References
-
- Fitzmaurice C., Allen C., Barber R.M., Barregard L., Bhutta Z.A., Brenner H., Dicker D.J., Chimed-Orchir O., Dandona R., Dandona L., et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived with Disability, and Disability-Adjusted Life-Years for 32 Cancer Groups, 1990 to 2015: A Systematic Analysis for the Global Burden of Disease Study. JAMA Oncol. 2017;3:524–548. doi: 10.1001/jamaoncol.2016.5688. - DOI - PMC - PubMed
-
- Kocarnik J.M., Compton K., Dean F.E., Fu W., Gaw B.L., Harvey J.D., Henrikson H.J., Lu D., Pennini A., Xu R., et al. Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life Years for 29 Cancer Groups from 2010 to 2019. JAMA Oncol. 2022;8:420–444. doi: 10.1001/jamaoncol.2021.6987. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

