Mitoxantrone and Mitoxantrone-Loaded Iron Oxide Nanoparticles Induce Cell Death in Human Pancreatic Ductal Adenocarcinoma Cell Spheroids
- PMID: 37049199
- PMCID: PMC10096321
- DOI: 10.3390/ma16072906
Mitoxantrone and Mitoxantrone-Loaded Iron Oxide Nanoparticles Induce Cell Death in Human Pancreatic Ductal Adenocarcinoma Cell Spheroids
Abstract
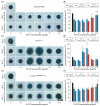
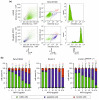
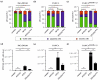
Pancreatic ductal adenocarcinoma is a hard-to-treat, deadly malignancy. Traditional treatments, such as surgery, radiation and chemotherapy, unfortunately are still not able to significantly improve long-term survival. Three-dimensional (3D) cell cultures might be a platform to study new drug types in a highly reproducible, resource-saving model within a relevant pathophysiological cellular microenvironment. We used a 3D culture of human pancreatic ductal adenocarcinoma cell lines to investigate a potential new treatment approach using superparamagnetic iron oxide nanoparticles (SPIONs) as a drug delivery system for mitoxantrone (MTO), a chemotherapeutic agent. We established a PaCa DD183 cell line and generated PANC-1SMAD4 (-/-) cells by using the CRISPR-Cas9 system, differing in a prognostically relevant mutation in the TGF-β pathway. Afterwards, we formed spheroids using PaCa DD183, PANC-1 and PANC-1SMAD4 (-/-) cells, and analyzed the uptake and cytotoxic effect of free MTO and MTO-loaded SPIONs by microscopy and flow cytometry. MTO and SPION-MTO-induced cell death in all tumor spheroids in a dose-dependent manner. Interestingly, spheroids with a SMAD4 mutation showed an increased uptake of MTO and SPION-MTO, while at the same time being more resistant to the cytotoxic effects of the chemotherapeutic agents. MTO-loaded SPIONs, with their ability for magnetic drug targeting, could be a future approach for treating pancreatic ductal adenocarcinomas.
Keywords: 3D cell culture; magnetic drug targeting; nanomedicine; pancreatic ductal adenocarcinoma; superparamagnetic iron oxide nanoparticles.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Treatment Efficiency of Free and Nanoparticle-Loaded Mitoxantrone for Magnetic Drug Targeting in Multicellular Tumor Spheroids.Molecules. 2015 Sep 30;20(10):18016-30. doi: 10.3390/molecules201018016. Molecules. 2015. PMID: 26437393 Free PMC article.
-
Toxicity of Mitoxantrone-loaded Superparamagnetic Iron Oxide Nanoparticles in a HT-29 Tumour Spheroid Model.Anticancer Res. 2016 Jun;36(6):3093-101. Anticancer Res. 2016. PMID: 27272833
-
Mitoxantrone-loaded superparamagnetic iron oxide nanoparticles as drug carriers for cancer therapy: Uptake and toxicity in primary human tubular epithelial cells.Nanotoxicology. 2016;10(5):557-66. doi: 10.3109/17435390.2015.1095364. Epub 2015 Oct 15. Nanotoxicology. 2016. PMID: 26468004
-
Superparamagnetic Iron Oxide Nanoparticles Carrying Chemotherapeutics Improve Drug Efficacy in Monolayer and Spheroid Cell Culture by Enabling Active Accumulation.Nanomaterials (Basel). 2020 Aug 11;10(8):1577. doi: 10.3390/nano10081577. Nanomaterials (Basel). 2020. PMID: 32796757 Free PMC article.
-
Intracellular Quantification and Localization of Label-Free Iron Oxide Nanoparticles by Holotomographic Microscopy.Nanotechnol Sci Appl. 2020 Dec 9;13:119-130. doi: 10.2147/NSA.S282204. eCollection 2020. Nanotechnol Sci Appl. 2020. PMID: 33328727 Free PMC article.
Cited by
-
CRISPR/Cas9 systems: Delivery technologies and biomedical applications.Asian J Pharm Sci. 2023 Nov;18(6):100854. doi: 10.1016/j.ajps.2023.100854. Epub 2023 Oct 21. Asian J Pharm Sci. 2023. PMID: 38089835 Free PMC article. Review.
-
Synergistic Effect of Chemotherapy and Magnetomechanical Actuation of Fe-Cr-Nb-B Magnetic Particles on Cancer Cells.ACS Omega. 2024 Jul 1;9(28):30518-30533. doi: 10.1021/acsomega.4c02189. eCollection 2024 Jul 16. ACS Omega. 2024. PMID: 39035922 Free PMC article.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

