The Impact of Obesity on the Excretion of Steroid Metabolites in Boys and Girls: A Comparison with Normal-Weight Children
- PMID: 37049573
- PMCID: PMC10097123
- DOI: 10.3390/nu15071734
The Impact of Obesity on the Excretion of Steroid Metabolites in Boys and Girls: A Comparison with Normal-Weight Children
Abstract
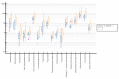
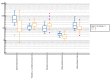
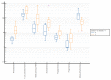
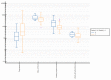
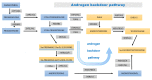
Obesity in childhood is associated with several steroid changes, which result from excess body mass. The aim of this study was to evaluate steroid metabolism in children with obesity compared with those with normal weight, especially in relation to sex and puberty progress. We analyzed the clinical data of 191 children, aged between 5 and 18 years, with 115 affected (64 girls and 51 boys) and 76 unaffected (35 girls and 41 boys) by obesity. Routine clinical assessment and pubertal stage evaluation based upon Tanner's scale were performed. In addition, to evaluate the impact of puberty, children with pre-adolescence and advanced puberty were divided into separate subgroups. Then, 24 h urine steroid excretion profiles were analyzed by gas chromatography/mass spectrometry. Significant differences in the excretion of steroid metabolites were found between normal weight children and children with obesity, especially in the prepubertal cohort. In this group, we observed enhanced activity in all the pathways of adrenal steroidogenesis. Raised excretion of mineralocorticoid derivatives such as tetrahydro-11-deoxycorticosterone, tetrahydrocorticosterone, and 5α-tetrahydrocorticosterone supported increased activity of this track. No significant differences were detected in the excreted free forms of cortisol and cortisone, while the excretion of their characteristic tetrahydro-derivatives was different. In pre-adolescent children with obesity, α-cortol and especially α-cortolone appeared to be excreted more abundantly than β-cortol or β-cortolone. Furthermore, in children with obesity, we observed elevated androgen excretion with an enhanced backdoor pathway. As puberty progressed, remarkable reduction in the differences between adolescents with and without obesity was demonstrated.
Keywords: adolescence; biomarkers; boys; childhood; girls; nutrition; obesity; puberty; urinary steroid metabolites.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationship that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Altered cortisol metabolism in polycystic ovary syndrome: insulin enhances 5alpha-reduction but not the elevated adrenal steroid production rates.J Clin Endocrinol Metab. 2003 Dec;88(12):5907-13. doi: 10.1210/jc.2003-030240. J Clin Endocrinol Metab. 2003. PMID: 14671189
-
Assessment of steroid enzymes action in children and adolescents with obesity.Steroids. 2023 Dec;200:109325. doi: 10.1016/j.steroids.2023.109325. Epub 2023 Oct 6. Steroids. 2023. PMID: 37806604
-
Assessment of circulating sex steroid levels in prepubertal and pubertal boys and girls by a novel ultrasensitive gas chromatography-tandem mass spectrometry method.J Clin Endocrinol Metab. 2010 Jan;95(1):82-92. doi: 10.1210/jc.2009-1140. Epub 2009 Nov 20. J Clin Endocrinol Metab. 2010. PMID: 19933393
-
Obesity and growth during childhood and puberty.World Rev Nutr Diet. 2013;106:135-41. doi: 10.1159/000342545. Epub 2013 Feb 11. World Rev Nutr Diet. 2013. PMID: 23428692 Review.
-
Linear growth and puberty in childhood obesity: what is new?Minerva Pediatr (Torino). 2021 Dec;73(6):563-571. doi: 10.23736/S2724-5276.21.06543-5. Epub 2021 Jul 26. Minerva Pediatr (Torino). 2021. PMID: 34309346 Review.
Cited by
-
Integrating Metabolomics and Gut Microbiota to Identify Key Biomarkers and Regulatory Pathways Underlying Metabolic Heterogeneity in Childhood Obesity.Nutrients. 2025 May 30;17(11):1876. doi: 10.3390/nu17111876. Nutrients. 2025. PMID: 40507148 Free PMC article.
-
Effects of Kimchi Intake on the Gut Microbiota and Metabolite Profiles of High-Fat-Induced Obese Rats.Nutrients. 2024 Sep 13;16(18):3095. doi: 10.3390/nu16183095. Nutrients. 2024. PMID: 39339693 Free PMC article.
-
Sex hormones, sex chromosomes, and microbiota: Identification of Akkermansia muciniphila as an estrogen-responsive microbiota.Microbiota Host. 2023;1(1):e230010. doi: 10.1530/mah-23-0010. Epub 2023 Oct 9. Microbiota Host. 2023. PMID: 37937163 Free PMC article.
References
-
- NCD Risk Factor Collaboration (NCD-RisC) Worldwide trends in body-mass index, underweight, overweight, and obesity from 1975 to 2016: A pooled analysis of 2416 population-based measurement studies in 128·9 million children, adolescents, and adults. Lancet. 2017;390:2627–2642. doi: 10.1016/S0140-6736(17)32129-3. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

