Fermented Aronia melanocarpa Inhibits Melanogenesis through Dual Mechanisms of the PI3K/AKT/GSK-3β and PKA/CREB Pathways
- PMID: 37049743
- PMCID: PMC10095632
- DOI: 10.3390/molecules28072981
Fermented Aronia melanocarpa Inhibits Melanogenesis through Dual Mechanisms of the PI3K/AKT/GSK-3β and PKA/CREB Pathways
Abstract
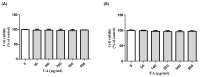
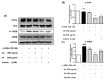
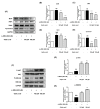
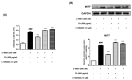
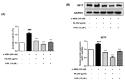
UV light causes excessive oxidative stress and abnormal melanin synthesis, which results in skin hyperpigmentation disorders such as freckles, sunspots, and age spots. Much research has been carried out to discover natural plants for ameliorating these disorders. Aronia melanocarpa contains various polyphenolic compounds with antioxidative activities, but its effects on melanogenesis have not been fully elucidated. In this study, we investigated the inhibitory effect of fermented Aronia melanocarpa (FA) fermented with Monascus purpureus on melanogenesis and its underlying mechanism in the B16F10 melanoma cell line. Our results indicate that FA inhibited tyrosinase activity and melanogenesis in alpha-melanocyte-stimulating hormone (α-MSH)-induced B16F10 cells. FA significantly downregulated the PKA/CREB pathway, resulting in decreased protein levels of tyrosinase, TRP-1, and MITF. FA also inhibited the transcription of MITF by increasing the phosphorylation levels of both GSK3β and AKT. Interestingly, we demonstrated that these results were owing to the significant increase in gallic acid, a phenolic compound of Aronia melanocarpa produced after the fermentation of Monascus purpureus. Taken together, our research suggests that Aronia melanocarpa fermented with Monascus purpureus acts as a melanin inhibitor and can be used as a potential cosmetic or therapeutic for improving hyperpigmentation disorders.
Keywords: Aronia melanocarpa; Monascus purpureus; anti-melanogenesis; fermentation; gallic acid; melanin.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Inhibitory Effect of Dried Pomegranate Concentration Powder on Melanogenesis in B16F10 Melanoma Cells; Involvement of p38 and PKA Signaling Pathways.Int J Mol Sci. 2015 Oct 13;16(10):24219-42. doi: 10.3390/ijms161024219. Int J Mol Sci. 2015. PMID: 26473849 Free PMC article.
-
Decursin prevents melanogenesis by suppressing MITF expression through the regulation of PKA/CREB, MAPKs, and PI3K/Akt/GSK-3β cascades.Biomed Pharmacother. 2022 Mar;147:112651. doi: 10.1016/j.biopha.2022.112651. Epub 2022 Jan 18. Biomed Pharmacother. 2022. PMID: 35063859
-
Mansonone E from Mansonia gagei Inhibited α-MSH-Induced Melanogenesis in B16 Cells by Inhibiting CREB Expression and Phosphorylation in the PI3K/Akt Pathway.Biol Pharm Bull. 2018;41(5):770-776. doi: 10.1248/bpb.b17-01045. Biol Pharm Bull. 2018. PMID: 29709914
-
A Comprehensive Review of the Functional Potential and Sustainable Applications of Aronia melanocarpa in the Food Industry.Plants (Basel). 2024 Dec 20;13(24):3557. doi: 10.3390/plants13243557. Plants (Basel). 2024. PMID: 39771257 Free PMC article. Review.
-
A Comprehensive Study to Explore Tyrosinase Inhibitory Medicinal Plants and Respective Phytochemicals for Hyperpigmentation; Molecular Approach and Future Perspectives.Curr Pharm Biotechnol. 2023;24(6):780-813. doi: 10.2174/1389201023666220823144242. Curr Pharm Biotechnol. 2023. PMID: 36017830 Review.
Cited by
-
The Pathogenesis of Oxidative Stress-Induced Chloasma and Its Therapeutic Implications.Clin Cosmet Investig Dermatol. 2025 Apr 16;18:929-947. doi: 10.2147/CCID.S520625. eCollection 2025. Clin Cosmet Investig Dermatol. 2025. PMID: 40255635 Free PMC article. Review.
-
Inhibitory Effects of Fermented Sprouted Oat Extracts on Oxidative Stress and Melanin Overproduction.Antioxidants (Basel). 2024 Apr 29;13(5):544. doi: 10.3390/antiox13050544. Antioxidants (Basel). 2024. PMID: 38790649 Free PMC article.
-
Rifampicin Repurposing Reveals Anti-Melanogenic Activity in B16F10 Melanoma Cells.Molecules. 2025 Feb 15;30(4):900. doi: 10.3390/molecules30040900. Molecules. 2025. PMID: 40005210 Free PMC article.
-
Functional Activities and Mechanisms of Aronia melanocarpa in Our Health.Curr Issues Mol Biol. 2024 Jul 26;46(8):8071-8087. doi: 10.3390/cimb46080477. Curr Issues Mol Biol. 2024. PMID: 39194694 Free PMC article. Review.
-
Biogenic Synthesis of Silver Nanoparticles Mediated by Aronia melanocarpa and Their Biological Evaluation.Life (Basel). 2024 Sep 23;14(9):1211. doi: 10.3390/life14091211. Life (Basel). 2024. PMID: 39337993 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

