Comparative assessment of the effects of DREADDs and endogenously expressed GPCRs in hippocampal astrocytes on synaptic activity and memory
- PMID: 37051110
- PMCID: PMC10083367
- DOI: 10.3389/fncel.2023.1159756
Comparative assessment of the effects of DREADDs and endogenously expressed GPCRs in hippocampal astrocytes on synaptic activity and memory
Abstract
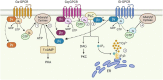
Designer Receptors Exclusively Activated by Designer Drugs (DREADDs) have proven themselves as one of the key in vivo techniques of modern neuroscience, allowing for unprecedented access to cellular manipulations in living animals. With respect to astrocyte research, DREADDs have become a popular method to examine the functional aspects of astrocyte activity, particularly G-protein coupled receptor (GPCR)-mediated intracellular calcium (Ca2+) and cyclic adenosine monophosphate (cAMP) dynamics. With this method it has become possible to directly link the physiological aspects of astrocytic function to cognitive processes such as memory. As a result, a multitude of studies have explored the impact of DREADD activation in astrocytes on synaptic activity and memory. However, the emergence of varying results prompts us to reconsider the degree to which DREADDs expressed in astrocytes accurately mimic endogenous GPCR activity. Here we compare the major downstream signaling mechanisms, synaptic, and behavioral effects of stimulating Gq-, Gs-, and Gi-DREADDs in hippocampal astrocytes of adult mice to those of endogenously expressed GPCRs.
Keywords: behavior; glia; signal transduction; signaling pathways; tripartite synapse.
Copyright © 2023 Lee, Mak and Verheijen.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Side-by-side comparison of the effects of Gq- and Gi-DREADD-mediated astrocyte modulation on intracellular calcium dynamics and synaptic plasticity in the hippocampal CA1.Mol Brain. 2021 Sep 20;14(1):144. doi: 10.1186/s13041-021-00856-w. Mol Brain. 2021. PMID: 34544455 Free PMC article.
-
Chemogenetic stimulation of the Gi pathway in astrocytes suppresses neuroinflammation.Pharmacol Res Perspect. 2021 Dec;9(6):e00822. doi: 10.1002/prp2.822. Pharmacol Res Perspect. 2021. PMID: 34676988 Free PMC article.
-
Gi/o protein-coupled receptors inhibit neurons but activate astrocytes and stimulate gliotransmission.Glia. 2019 Jun;67(6):1076-1093. doi: 10.1002/glia.23589. Epub 2019 Feb 23. Glia. 2019. PMID: 30801845 Free PMC article.
-
Chemogenetic manipulation of astrocyte activity at the synapse- a gateway to manage brain disease.Front Cell Dev Biol. 2023 Jul 18;11:1193130. doi: 10.3389/fcell.2023.1193130. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37534103 Free PMC article. Review.
-
The Use of DREADDs to Deconstruct Behavior.Front Genet. 2016 May 17;7:70. doi: 10.3389/fgene.2016.00070. eCollection 2016. Front Genet. 2016. PMID: 27242888 Free PMC article. Review.
Cited by
-
Adrenergic signaling gates astrocyte responsiveness to neurotransmitters and control of neuronal activity.bioRxiv [Preprint]. 2024 Sep 23:2024.09.23.614537. doi: 10.1101/2024.09.23.614537. bioRxiv. 2024. Update in: Science. 2025 May 15;388(6748):763-768. doi: 10.1126/science.adq5729. PMID: 39386551 Free PMC article. Updated. Preprint.
-
Astrocytic Regulation of Endocannabinoid-Dependent Synaptic Plasticity in the Dorsolateral Striatum.Int J Mol Sci. 2024 Jan 1;25(1):581. doi: 10.3390/ijms25010581. Int J Mol Sci. 2024. PMID: 38203752 Free PMC article.
-
Retrotrapezoid nucleus chemoreception: mechanisms of function and contributions to disordered breathing in disease.Trends Neurosci. 2025 Aug 15:S0166-2236(25)00148-1. doi: 10.1016/j.tins.2025.07.006. Online ahead of print. Trends Neurosci. 2025. PMID: 40816914 Review.
-
Astrocytes in Rodent Anxiety-Related Behavior: Role of Calcium and Beyond.Int J Mol Sci. 2025 Mar 19;26(6):2774. doi: 10.3390/ijms26062774. Int J Mol Sci. 2025. PMID: 40141416 Free PMC article. Review.
-
Astrocytes control recent and remote memory strength by affecting the recruitment of the CA1→ACC projection to engrams.Cell Rep. 2024 Mar 26;43(3):113943. doi: 10.1016/j.celrep.2024.113943. Epub 2024 Mar 13. Cell Rep. 2024. PMID: 38483907 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

