Complement lectin pathway activation is associated with COVID-19 disease severity, independent of MBL2 genotype subgroups
- PMID: 37051252
- PMCID: PMC10084477
- DOI: 10.3389/fimmu.2023.1162171
Complement lectin pathway activation is associated with COVID-19 disease severity, independent of MBL2 genotype subgroups
Abstract
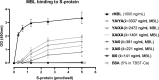
Introduction: While complement is a contributor to disease severity in severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infections, all three complement pathways might be activated by the virus. Lectin pathway activation occurs through different pattern recognition molecules, including mannan binding lectin (MBL), a protein shown to interact with SARS-CoV-2 proteins. However, the exact role of lectin pathway activation and its key pattern recognition molecule MBL in COVID-19 is still not fully understood.
Methods: We therefore investigated activation of the lectin pathway in two independent cohorts of SARS-CoV-2 infected patients, while also analysing MBL protein levels and potential effects of the six major single nucleotide polymorphisms (SNPs) found in the MBL2 gene on COVID-19 severity and outcome.
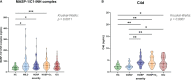
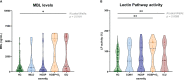
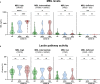
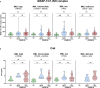
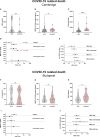
Results: We show that the lectin pathway is activated in acute COVID-19, indicated by the correlation between complement activation product levels of the MASP-1/C1-INH complex (p=0.0011) and C4d (p<0.0001) and COVID-19 severity. Despite this, genetic variations in MBL2 are not associated with susceptibility to SARS-CoV-2 infection or disease outcomes such as mortality and the development of Long COVID.
Conclusion: In conclusion, activation of the MBL-LP only plays a minor role in COVID-19 pathogenesis, since no clinically meaningful, consistent associations with disease outcomes were noted.
Keywords: COVID-19; MBL2 genotypes; lectin pathway activation; lectin pathway of complement; mannose binding lectin (MBL); severe acute respiratory coronavirus 2 (SARS-CoV-2).
Copyright © 2023 Hurler, Szilágyi, Mescia, Bergamaschi, Mező, Sinkovits, Réti, Müller, Iványi, Gál, Gopcsa, Reményi, Szathmáry, Lakatos, Szlávik, Bobek, Prohászka, Förhécz, Csuka, Kajdácsi, Cervenak, Kiszel, Masszi, Vályi-Nagy, Würzner, Cambridge Institute of Therapeutic Immunology and Infectious Disease-National Institute of Health Research (CITIID-NIHR) COVID BioResource Collaboration, Lyons, Toonen and Prohászka.
Conflict of interest statement
Author ET is an employee of Hycult Biotech. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Association between mannose binding lectin gene polymorphisms and clinical severity of COVID-19 in children.Mol Biol Rep. 2023 Jul;50(7):5871-5877. doi: 10.1007/s11033-023-08524-z. Epub 2023 May 25. Mol Biol Rep. 2023. PMID: 37231213 Free PMC article.
-
A likely association between low mannan-binding lectin level and brain fog onset in long COVID patients.Front Immunol. 2023 Jun 16;14:1191083. doi: 10.3389/fimmu.2023.1191083. eCollection 2023. Front Immunol. 2023. PMID: 37398656 Free PMC article.
-
The MBL2 genotype relates to COVID-19 severity and may help to select the optimal therapy.Clin Chem Lab Med. 2023 Jun 15;61(12):2143-2149. doi: 10.1515/cclm-2023-0183. Print 2023 Nov 27. Clin Chem Lab Med. 2023. PMID: 37313996
-
Status of mannose-binding lectin (MBL) and complement system in COVID-19 patients and therapeutic applications of antiviral plant MBLs.Mol Cell Biochem. 2021 Aug;476(8):2917-2942. doi: 10.1007/s11010-021-04107-3. Epub 2021 Mar 21. Mol Cell Biochem. 2021. PMID: 33745077 Free PMC article. Review.
-
The Influence of the Lectin Pathway of Complement Activation on Infections of the Respiratory System.Front Immunol. 2020 Oct 21;11:585243. doi: 10.3389/fimmu.2020.585243. eCollection 2020. Front Immunol. 2020. PMID: 33193407 Free PMC article. Review.
Cited by
-
The Lectin Pathway of the Complement System-Activation, Regulation, Disease Connections and Interplay with Other (Proteolytic) Systems.Int J Mol Sci. 2024 Jan 26;25(3):1566. doi: 10.3390/ijms25031566. Int J Mol Sci. 2024. PMID: 38338844 Free PMC article. Review.
-
Control of complement-induced inflammatory responses to SARS-CoV-2 infection by anti-SARS-CoV-2 antibodies.EMBO J. 2024 Apr;43(7):1135-1163. doi: 10.1038/s44318-024-00061-0. Epub 2024 Feb 28. EMBO J. 2024. PMID: 38418557 Free PMC article.
-
Anti-Inflammatory Cytokine Profiles in Thrombotic Thrombocytopenic Purpura-Differences Compared to COVID-19.Int J Mol Sci. 2024 Sep 17;25(18):10007. doi: 10.3390/ijms251810007. Int J Mol Sci. 2024. PMID: 39337495 Free PMC article.
-
sMR and PTX3 levels associate with COVID-19 outcome and survival but not with Long COVID.iScience. 2024 Jun 1;27(7):110162. doi: 10.1016/j.isci.2024.110162. eCollection 2024 Jul 19. iScience. 2024. PMID: 39027374 Free PMC article.
-
Emerging role of complement in COVID-19 and other respiratory virus diseases.Cell Mol Life Sci. 2024 Feb 18;81(1):94. doi: 10.1007/s00018-024-05157-8. Cell Mol Life Sci. 2024. PMID: 38368584 Free PMC article. Review.
References
-
- World Health Organization. COVID-19 weekly epidemiological update, 128. WHO, World Health Organization; (2023). Available at: https://apps.who.int/iris/handle/10665/365807.
-
- Ji X, Olinger GG, Aris S, Chen Y, Gewurz H, Spear GT. Mannose-binding lectin binds to Ebola and marburg envelope glycoproteins, resulting in blocking of virus interaction with DC-SIGN and complement-mediated virus neutralization. J Gen Virol (2005) 86(Pt 9):2535–42. doi: 10.1099/vir.0.81199-0 - DOI - PubMed
-
- Mak TW, Saunders ME, Jett BD. eds. Chapter 3 - innate immunity. In: Primer to the immune response. Academic Cell. 2014, 55-83. doi: 10.1016/B978-0-12-385245-8.00003-0 - DOI
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

