Nose-to-brain delivery of self-assembled curcumin-lactoferrin nanoparticles: Characterization, neuroprotective effect and in vivo pharmacokinetic study
- PMID: 37051277
- PMCID: PMC10084992
- DOI: 10.3389/fbioe.2023.1168408
Nose-to-brain delivery of self-assembled curcumin-lactoferrin nanoparticles: Characterization, neuroprotective effect and in vivo pharmacokinetic study
Abstract
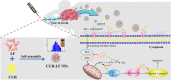
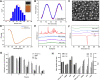
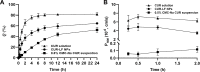
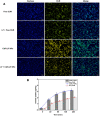
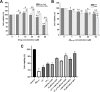
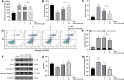
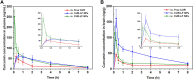
Curcumin (CUR) is a natural polyphenol extract with significant antioxidant and anti-inflammatory effects, which indicates its great potential for neuroprotection. Lactoferrin (LF), a commonly used oral carrier and targeting ligand, has not been reported as a multifunctional nanocarrier for nose-to-brain delivery. This study aims to develop a nose-to-brain delivery system of curcumin-lactoferrin nanoparticles (CUR-LF NPs) and to further evaluate the neuroprotective effects in vitro and brain accumulation in vivo. Herein, CUR-LF NPs were prepared by the desolvation method with a particle size of 84.8 ± 6.5 nm and a zeta potential of +22.8 ± 4.3 mV. The permeability coefficient of CUR-LF NPs (4.36 ± 0.79 × 10-6 cm/s) was 50 times higher than that of CUR suspension (0.09 ± 0.04 × 10-6 cm/s) on MDCK monolayer, indicating that the nanoparticles could improve the absorption efficiency of CUR in the nasal cavity. Moreover, CUR-LF NPs showed excellent protection against Aβ25-35-induced nerve damage in PC12 cells. In vivo pharmacokinetic studies showed that the brain-targeting efficiency of CUR-LF NPs via IN administration was 248.1%, and the nose-to-brain direct transport percentage was 59.7%. Collectively, nose-to-brain delivery of CUR-LF NPs is capable of achieving superior brain enrichment and potential neuroprotective effects.
Keywords: Aβ25-35; curcumin-lactoferrin nanoparticles; neuroprotective effect; nose-to-brain; pharmacokinetic.
Copyright © 2023 Li, Tan, Zhang, Cheng, Liu, Li and Hou.
Conflict of interest statement
LT, YC, and YL were employed by Sichuan Purity Pharmaceutical Co., Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Intranasal delivery of Huperzine A to the brain using lactoferrin-conjugated N-trimethylated chitosan surface-modified PLGA nanoparticles for treatment of Alzheimer's disease.Int J Nanomedicine. 2018 Feb 1;13:705-718. doi: 10.2147/IJN.S151474. eCollection 2018. Int J Nanomedicine. 2018. PMID: 29440896 Free PMC article.
-
Folic acid-modified lactoferrin nanoparticles coated with a laminarin layer loaded curcumin with dual-targeting for ulcerative colitis treatment.Int J Biol Macromol. 2023 Mar 31;232:123229. doi: 10.1016/j.ijbiomac.2023.123229. Epub 2023 Jan 13. Int J Biol Macromol. 2023. PMID: 36642354
-
Lactoferrin-modified rotigotine nanoparticles for enhanced nose-to-brain delivery: LESA-MS/MS-based drug biodistribution, pharmacodynamics, and neuroprotective effects.Int J Nanomedicine. 2018 Jan 9;13:273-281. doi: 10.2147/IJN.S151475. eCollection 2018. Int J Nanomedicine. 2018. PMID: 29391788 Free PMC article.
-
Formulation of lactoferrin decorated dextran based chitosan-coated europium metal-organic framework for targeted delivery of curcumin.Int J Biol Macromol. 2024 Feb;259(Pt 2):129325. doi: 10.1016/j.ijbiomac.2024.129325. Epub 2024 Jan 12. Int J Biol Macromol. 2024. PMID: 38219935
-
Lactoferrin as a Versatile Agent in Nanoparticle Applications: From Therapeutics to Agriculture.Nanomaterials (Basel). 2024 Dec 16;14(24):2018. doi: 10.3390/nano14242018. Nanomaterials (Basel). 2024. PMID: 39728554 Free PMC article. Review.
Cited by
-
Research progress in brain-targeted nasal drug delivery.Front Aging Neurosci. 2024 Jan 17;15:1341295. doi: 10.3389/fnagi.2023.1341295. eCollection 2023. Front Aging Neurosci. 2024. PMID: 38298925 Free PMC article. Review.
-
In Vitro and In Vivo Evaluation of Lactoferrin-Modified Liposomal Etomidate with Enhanced Brain-Targeting Effect for General Anesthesia.Pharmaceutics. 2024 Jun 14;16(6):805. doi: 10.3390/pharmaceutics16060805. Pharmaceutics. 2024. PMID: 38931926 Free PMC article.
-
Overcoming Biopotency Barriers: Advanced Oral Delivery Strategies for Enhancing the Efficacy of Bioactive Food Ingredients.Adv Sci (Weinh). 2024 Nov;11(44):e2401172. doi: 10.1002/advs.202401172. Epub 2024 Oct 3. Adv Sci (Weinh). 2024. PMID: 39361948 Free PMC article. Review.
-
A Comprehensive Review of Nanoparticles: From Classification to Application and Toxicity.Molecules. 2024 Jul 25;29(15):3482. doi: 10.3390/molecules29153482. Molecules. 2024. PMID: 39124888 Free PMC article. Review.
-
KLVFF Conjugated Curcumin Microemulsion-Based Hydrogel for Transnasal Route: Formulation Development, Optimization, Physicochemical Characterization, and Ex Vivo Evaluation.Gels. 2023 Jul 28;9(8):610. doi: 10.3390/gels9080610. Gels. 2023. PMID: 37623065 Free PMC article.
References
LinkOut - more resources
Full Text Sources

