Characterization of the Molecular Weight of Hyaluronan in Eye Products Using a Novel Method of Size Exclusion High-Pressure Liquid Chromatography
- PMID: 37052911
- PMCID: PMC10103721
- DOI: 10.1167/tvst.12.4.13
Characterization of the Molecular Weight of Hyaluronan in Eye Products Using a Novel Method of Size Exclusion High-Pressure Liquid Chromatography
Abstract
Purpose: Hyaluronan (HA) exists in two forms, high molecular weight HA (HMWHA) and low molecular weight HA (LMWHA), which have distinct physiological functions. Therefore it is imperative to know the form of HA within pharmaceutical products, including eye products. This study developed an accurate, sensitive, and quantitative method to characterize the form of HA in eye products. Thereafter, the effects of the HA-containing eye products on corneal wound healing were investigated.
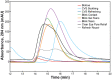
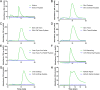
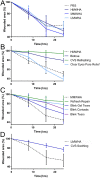
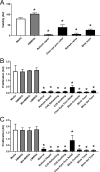
Methods: The MW distributions and concentrations of HA in over the counter eye products were determined by size exclusion chromatography (SEC) high-pressure liquid chromatography (HPLC). The effects of the eye products containing HA on corneal wound healing were characterized both in vitro and in vivo using the scratch assay and the debridement wound model, respectively.
Results: The concentrations and MWs of HA were successfully determined within a range of 0.014 to 0.25 mg/mL using SEC HPLC. The concentrations of HA in the ophthalmic products varied from 0.14 to 4.0 mg/mL and the MWs varied from ∼100 kDa to >2500 kDa. All but one HA-containing eye product had an inhibitory effect on corneal wound healing, whereas pure HA promoted corneal wound healing.
Conclusions: A novel SEC-HPLC method was developed for quantifying and characterizing the MW of HA in eye products. Although HA promoted corneal wound healing, HA-containing eye products inhibited corneal wound healing, likely caused by preservatives.
Translational relevance: SEC-HPLC could be implemented as a routine method for determining the form of HA in commercially available ophthalmic products.
Conflict of interest statement
Disclosure:
Figures



Similar articles
-
ROS-Mediated Fragmentation Alters the Effects of Hyaluronan on Corneal Epithelial Wound Healing.Biomolecules. 2023 Sep 13;13(9):1385. doi: 10.3390/biom13091385. Biomolecules. 2023. PMID: 37759785 Free PMC article.
-
Impact of Hyaluronic Acid-Containing Artificial Tear Products on Reepithelialization in an In Vivo Corneal Wound Model.J Ocul Pharmacol Ther. 2018 May;34(4):360-364. doi: 10.1089/jop.2017.0080. Epub 2018 Feb 2. J Ocul Pharmacol Ther. 2018. PMID: 29394128 Free PMC article.
-
Comparative studies of hyaluronan in marketed ophthalmic products.Optom Vis Sci. 2014 Jan;91(1):32-8. doi: 10.1097/OPX.0000000000000100. Optom Vis Sci. 2014. PMID: 24366432
-
Hyaluronan as an ophthalmic viscoelastic device.Curr Pharm Biotechnol. 2008 Aug;9(4):236-8. doi: 10.2174/138920108785161596. Curr Pharm Biotechnol. 2008. PMID: 18691081 Review.
-
HA metabolism in skin homeostasis and inflammatory disease.Food Chem Toxicol. 2017 Mar;101:128-138. doi: 10.1016/j.fct.2017.01.012. Epub 2017 Jan 18. Food Chem Toxicol. 2017. PMID: 28109952 Review.
Cited by
-
ROS-Mediated Fragmentation Alters the Effects of Hyaluronan on Corneal Epithelial Wound Healing.Biomolecules. 2023 Sep 13;13(9):1385. doi: 10.3390/biom13091385. Biomolecules. 2023. PMID: 37759785 Free PMC article.
-
Synthesis and Engineering of Hyaluronic Acid-Gelatin Hydrogels with Improved Cellular Attachment and Growth.Polymers (Basel). 2024 Dec 3;16(23):3410. doi: 10.3390/polym16233410. Polymers (Basel). 2024. PMID: 39684154 Free PMC article.
-
Hyaluronate Protects From Benzalkonium Chloride-Induced Ocular Surface Toxicity.Transl Vis Sci Technol. 2024 Oct 1;13(10):31. doi: 10.1167/tvst.13.10.31. Transl Vis Sci Technol. 2024. PMID: 39432403 Free PMC article.
-
Age related changes in hyaluronan expression leads to Meibomian gland dysfunction.Matrix Biol. 2023 Dec;124:23-38. doi: 10.1016/j.matbio.2023.11.002. Epub 2023 Nov 8. Matrix Biol. 2023. PMID: 37949327 Free PMC article.
-
Comparative Analysis of Age-Associated Changes in Meibum Composition, Distribution, and Function in Mice With Altered Hyaluronan Expression.Invest Ophthalmol Vis Sci. 2025 Jul 1;66(9):72. doi: 10.1167/iovs.66.9.72. Invest Ophthalmol Vis Sci. 2025. PMID: 40736174 Free PMC article.
References
-
- Lee JY, Spicer AP. Hyaluronan: A multifunctional, megaDalton, stealth molecule. Curr Opin Cell Biol. 2000; 12: 581–586. - PubMed
-
- Grigorij K, Ladislav Š, Robert S, Peter G. Hyaluronic acid: A natural biopolymer with a broad range of biomedical and industrial applications. Biotechnol Lett. 2007; 29: 17–25. - PubMed
-
- Gong H, Underhill CB, Freddo TF. Hyaluronan in the bovine ocular anterior segment, with emphasis on the outflow pathways. Invest Ophthalmol Vis Sci. 1994; 35: 4328–4332. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

