Synthetic symbiosis between a cyanobacterium and a ciliate toward novel chloroplast-like endosymbiosis
- PMID: 37055487
- PMCID: PMC10102011
- DOI: 10.1038/s41598-023-33321-w
Synthetic symbiosis between a cyanobacterium and a ciliate toward novel chloroplast-like endosymbiosis
Abstract
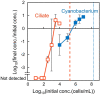
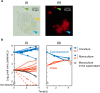
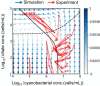
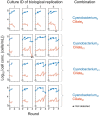
Chloroplasts are thought to have co-evolved through endosymbiosis, after a cyanobacterial-like prokaryote was engulfed by a eukaryotic cell; however, it is impossible to observe the process toward chloroplasts. In this study, we constructed an experimental symbiosis model to observe the initial stage in the process from independent organisms to a chloroplast-like organelle. Our system of synthetic symbiosis is capable of long-term coculture of two model organisms: a cyanobacterium (Synechocystis sp. PCC6803) as a symbiont and a ciliate (Tetrahymena thermophila) as a host with endocytic ability. The experimental system was clearly defined, because we used a synthetic medium and the cultures were shaken to avoid spatial complexity. We determined the experimental conditions for sustainable coculture, by analyzing population dynamics using a mathematical model. We experimentally demonstrated that the coculture was sustainable for at least 100 generations, through serial transfers. Moreover, we found that cells isolated after the serial transfer improved the probability of coexistence of both species without extinction in re-coculture. The constructed system will be useful for understanding the initial stage of primary endosymbiosis from cyanobacteria to chloroplasts, i.e., the origin of algae and plants.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

Similar articles
-
Complex origins of chloroplast membranes with photosynthetic machineries: multiple transfers of genes from divergent organisms at different times or a single endosymbiotic event?J Plant Res. 2020 Jan;133(1):15-33. doi: 10.1007/s10265-019-01157-z. Epub 2019 Dec 6. J Plant Res. 2020. PMID: 31811433 Free PMC article.
-
Are Cyanobacteria an Ancestor of Chloroplasts or Just One of the Gene Donors for Plants and Algae?Genes (Basel). 2021 May 27;12(6):823. doi: 10.3390/genes12060823. Genes (Basel). 2021. PMID: 34071987 Free PMC article.
-
Adaptation of a cyanobacterium to a biochemically rich environment in experimental evolution as an initial step toward a chloroplast-like state.PLoS One. 2014 May 29;9(5):e98337. doi: 10.1371/journal.pone.0098337. eCollection 2014. PLoS One. 2014. PMID: 24874568 Free PMC article.
-
Origin and evolution of the chloroplast division machinery.J Plant Res. 2005 Oct;118(5):295-306. doi: 10.1007/s10265-005-0226-2. Epub 2005 Sep 13. J Plant Res. 2005. PMID: 16143878 Review.
-
Primary Endosymbiosis: Emergence of the Primary Chloroplast and the Chromatophore, Two Independent Events.Methods Mol Biol. 2018;1829:3-16. doi: 10.1007/978-1-4939-8654-5_1. Methods Mol Biol. 2018. PMID: 29987711 Review.
Cited by
-
Biodiversity and Constrained Information Dynamics in Ecosystems: A Framework for Living Systems.Entropy (Basel). 2023 Dec 5;25(12):1624. doi: 10.3390/e25121624. Entropy (Basel). 2023. PMID: 38136504 Free PMC article.
-
Molecular and biochemical insights from natural and engineered photosynthetic endosymbiotic systems.Curr Opin Chem Biol. 2025 Aug;87:102598. doi: 10.1016/j.cbpa.2025.102598. Epub 2025 Apr 18. Curr Opin Chem Biol. 2025. PMID: 40252292 Free PMC article. Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

