Multiparametric magnetic resonance imaging for radiation therapy response monitoring in soft tissue sarcomas: a histology and MRI co-registration algorithm
- PMID: 37056570
- PMCID: PMC10086213
- DOI: 10.7150/thno.81938
Multiparametric magnetic resonance imaging for radiation therapy response monitoring in soft tissue sarcomas: a histology and MRI co-registration algorithm
Abstract
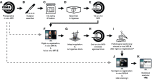
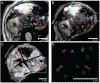
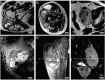
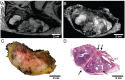
Rationale: To establish a spatially exact co-registration procedure between in vivo multiparametric magnetic resonance imaging (mpMRI) and (immuno)histopathology of soft tissue sarcomas (STS) to identify imaging parameters that reflect radiation therapy response of STS. Methods: The mpMRI-Protocol included diffusion-weighted (DWI), intravoxel-incoherent motion (IVIM), and dynamic contrast-enhancing (DCE) imaging. The resection specimen was embedded in 6.5% agarose after initial fixation in formalin. To ensure identical alignment of histopathological sectioning and in vivo imaging, an ex vivo MRI scan of the specimen was rigidly co-registered with the in vivo mpMRI. The deviating angulation of the specimen to the in vivo location of the tumor was determined. The agarose block was trimmed accordingly. A second ex vivo MRI in a dedicated localizer with a 4 mm grid was performed, which was matched to a custom-built sectioning machine. Microtomy sections were stained with hematoxylin and eosin. Immunohistochemical staining was performed with anti-ALDH1A1 antibodies as a radioresistance and anti-MIB1 antibodies as a proliferation marker. Fusion of the digitized microtomy sections with the in vivo mpMRI was accomplished through nonrigid co-registration to the in vivo mpMRI. Co-registration accuracy was qualitatively assessed by visual assessment and quantitatively evaluated by computing target registration errors (TRE). Results: The study sample comprised nine tumor sections from three STS patients. Visual assessment after nonrigid co-registration showed a strong morphological correlation of the histopathological specimens with ex vivo MRI and in vivo mpMRI after neoadjuvant radiation therapy. Quantitative assessment of the co-registration procedure using TRE analysis of different pairs of pathology and MRI sections revealed highly accurate structural alignment, with a total median TRE of 2.25 mm (histology - ex vivo MRI), 2.22 mm (histology - in vivo mpMRI), and 2.02 mm (ex vivo MRI - in vivo mpMRI). There was no significant difference between TREs of the different pairs of sections or caudal, middle, and cranial tumor parts, respectively. Conclusion: Our initial results show a promising approach to obtaining accurate co-registration between histopathology and in vivo MRI for STS. In a larger cohort of patients, the method established here will enable the prospective identification and validation of in vivo imaging biomarkers for radiation therapy response prediction and monitoring in STS patients via precise molecular and cellular correlation.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures



References
-
- Group EESNW. Soft tissue and visceral sarcomas: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2012;23(Suppl 7):vii92–9. - PubMed
-
- Ray-Coquard I, Serre D, Reichardt P, Martin-Broto J, Bauer S. Options for treating different soft tissue sarcoma subtypes. Future Oncol. 2018;14:25–49. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

