Magnetic Resonance Imaging Techniques in Peripheral Arterial Disease
- PMID: 37058352
- PMCID: PMC10468560
- DOI: 10.1089/wound.2022.0161
Magnetic Resonance Imaging Techniques in Peripheral Arterial Disease
Abstract
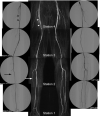
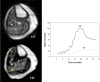
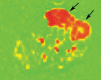
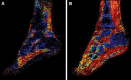
Significance: Peripheral arterial disease (PAD) leads to a significant burden of morbidity and impaired quality of life globally. Diabetes is a significant risk factor accelerating the development of PAD with an associated increase in the risk of chronic wounds, tissue, and limb loss. Various magnetic resonance imaging (MRI) techniques are being increasingly acknowledged as useful methods of accurately assessing PAD. Recent Advances: Conventionally utilized MRI techniques for assessing macrovascular disease have included contrast enhanced magnetic resonance angiography (MRA), noncontrast time of flight MRA, and phase contrast MRI, but have significant limitations. In recent years, novel noncontrast MRI methods assessing skeletal muscle perfusion and metabolism such as arterial spin labeling (ASL), blood-oxygen-level dependent (BOLD) imaging, and chemical exchange saturation transfer (CEST) have emerged. Critical Issues: Conventional non-MRI (such as ankle-brachial index, arterial duplex ultrasonography, and computed tomographic angiography) and MRI based modalities image the macrovasculature. The underlying mechanisms of PAD that result in clinical manifestations are, however, complex, and imaging modalities that can assess the interaction between impaired blood flow, microvascular tissue perfusion, and muscular metabolism are necessary. Future Directions: Further development and clinical validation of noncontrast MRI methods assessing skeletal muscle perfusion and metabolism, such as ASL, BOLD, CEST, intravoxel incoherent motion microperfusion, and techniques that assess plaque composition, are advancing this field. These modalities can provide useful prognostic data and help in reliable surveillance of outcomes after interventions.
Keywords: arterial spin labeling; blood-oxygen-level dependent imaging; dynamic contrast-enhanced imaging; noncontrast magnetic resonance angiography; peripheral arterial disease; quiescent-interval single-shot.
Conflict of interest statement
Christopher M Kramer is supported by a research grant from NHLBI in this area. No ghostwriting services were used.
Figures

Similar articles
-
Evaluation of skeletal muscle microvascular perfusion of lower extremities by cardiovascular magnetic resonance arterial spin labeling, blood oxygenation level-dependent, and intravoxel incoherent motion techniques.J Cardiovasc Magn Reson. 2018 Mar 19;20(1):18. doi: 10.1186/s12968-018-0441-3. J Cardiovasc Magn Reson. 2018. PMID: 29551091 Free PMC article.
-
Recent advances in magnetic resonance imaging for peripheral artery disease.Vasc Med. 2018 Apr;23(2):143-152. doi: 10.1177/1358863X18754694. Vasc Med. 2018. PMID: 29633922 Free PMC article. Review.
-
Accuracy of non-contrast quiescent-interval single-shot and quiescent-interval single-shot arterial spin-labelled magnetic resonance angiography in assessment of peripheral arterial disease in a diabetic population.J Med Imaging Radiat Oncol. 2020 Feb;64(1):35-43. doi: 10.1111/1754-9485.12987. J Med Imaging Radiat Oncol. 2020. PMID: 32043319
-
Arterial spin labeling MR imaging reproducibly measures peak-exercise calf muscle perfusion: a study in patients with peripheral arterial disease and healthy volunteers.JACC Cardiovasc Imaging. 2012 Dec;5(12):1224-30. doi: 10.1016/j.jcmg.2012.03.022. JACC Cardiovasc Imaging. 2012. PMID: 23236972 Free PMC article.
-
Noncontrast Magnetic Resonance Angiography for the Diagnosis of Peripheral Vascular Disease.Circ Cardiovasc Imaging. 2019 May;12(5):e008844. doi: 10.1161/CIRCIMAGING.118.008844. Circ Cardiovasc Imaging. 2019. PMID: 31088154 Review.
Cited by
-
Current Opinion on Diagnosis of Peripheral Artery Disease in Diabetic Patients.Medicina (Kaunas). 2024 Jul 20;60(7):1179. doi: 10.3390/medicina60071179. Medicina (Kaunas). 2024. PMID: 39064610 Free PMC article. Review.
-
Accuracy of 3D CE-MRA combined with ultrasound microbubble angiography for the diagnosis of lower extremity atherosclerotic occlusion.Am J Transl Res. 2025 Feb 15;17(2):818-831. doi: 10.62347/HVBK1487. eCollection 2025. Am J Transl Res. 2025. PMID: 40092093 Free PMC article.
-
Background suppression single-shot electrocardiogram trigger non-enhanced magnetic resonance angiography in lower extremity blood vessels: a comparative study.Quant Imaging Med Surg. 2025 Feb 1;15(2):1312-1323. doi: 10.21037/qims-24-1120. Epub 2025 Jan 22. Quant Imaging Med Surg. 2025. PMID: 39995725 Free PMC article.
References
-
- Greve JM, Williams SP, Bernstein LJ, et al. . Reactive hyperemia and BOLD MRI demonstrate that VEGF inhibition, age, and atherosclerosis adversely affect functional recovery in a murine model of peripheral artery disease. J Magn Reson Imaging 2008;28(4):996–1004; doi:10.1002/jmri.21517 - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

