Clinical management and diagnosis of CLN2 disease: consensus of the Brazilian experts group
- PMID: 37059438
- PMCID: PMC10104757
- DOI: 10.1055/s-0043-1761434
Clinical management and diagnosis of CLN2 disease: consensus of the Brazilian experts group
Abstract
Neuronal ceroid lipofuscinosis type 2 (CLN2) is a rare neurodegenerative genetic disease that affects children in early life. Its classic form is rapidly progressive, leading to death within the first 10 years. The urge for earlier diagnosis increases with the availability of enzyme replacement therapy. A panel of nine Brazilian child neurologists combined their expertise in CLN2 with evidence from the medical literature to establish a consensus to manage this disease in Brazil. They voted 92 questions including diagnosis, clinical manifestations, and treatment of the disease, considering the access to healthcare in this country. Clinicians should suspect CLN2 disease in any child, from 2 to 4 years old, with language delay and epilepsy. Even though the classic form is the most prevalent, atypical cases with different phenotypes can be found. Electroencephalogram, magnetic resonance imaging, molecular and biochemical testing are the main tools to investigate and confirm the diagnosis. However, we have limited access to molecular testing in Brazil, and rely on the support from the pharmaceutical industry. The management of CLN2 should involve a multidisciplinary team and focus on the quality of life of patients and on family support. Enzyme replacement therapy with Cerliponase α is an innovative treatment approved in Brazil since 2018; it delays functional decline and provides quality of life. Given the difficulties for the diagnosis and treatment of rare diseases in our public health system, the early diagnosis of CLN2 needs improvement as enzyme replacement therapy is available and modifies the prognosis of patients.
Lipofuscinose ceróide neuronal (CLN2) é uma doença genética neurodegenerativa rara que afeta crianças nos primeiros anos de vida. A sua forma clássica é rapidamente progressiva, levando à morte nos primeiros 10 anos. A necessidade de um diagnóstico precoce aumenta com a disponibilidade do tratamento de terapia enzimática. Um painel de nove neurologistas infantis brasileiros combinou sua experiência em CLN2 com evidências da literatura médica para estabelecer um consenso no manejo desta doença no Brasil. Eles votaram 92 questões abordando diagnóstico, manifestações clínicas e tratamento, considerando o acesso à saúde no Brasil. Deve-se suspeitar de CLN2 em qualquer criança de 2 a 4 anos de idade que apresente atraso de linguagem e epilepsia. Apesar da forma clássica ser a mais prevalente, podem ser encontrados casos atípicos com diferentes fenótipos. Eletroencefalograma, ressonância magnética, testes moleculares e bioquímicos são as principais ferramentas para investigar e confirmar o diagnóstico. No entanto, o acesso aos testes moleculares é limitado no Brasil, necessitando contar com o apoio da indústria farmacêutica. O manejo da CLN2 deve envolver uma equipe multidisciplinar e focar na qualidade de vida dos pacientes e no apoio familiar. A terapia de reposição enzimática com Cerliponase alfa é um tratamento inovador aprovado no Brasil desde 2018; ele retarda o declínio funcional e proporciona qualidade de vida. Diante das dificuldades para o diagnóstico e tratamento de doenças raras em nosso sistema público de saúde, o diagnóstico precoce de CLN2 precisa de melhorias pois a terapia de reposição enzimática está disponível e modifica o prognóstico dos pacientes.
Academia Brasileira de Neurologia. This is an open access article published by Thieme under the terms of the Creative Commons Attribution 4.0 International License, permitting copying and reproduction so long as the original work is given appropriate credit (https://creativecommons.org/licenses/by/4.0/).
Conflict of interest statement
AP: has received speaker honorarium and is a consultant at BioMarin; EKE: has received speaker honorarium from BioMarin and Janssen; FK: has received financial support from PTC Pharmaceuticals, Actelion-Janssen, and BioMarin for medical education programs. He is the medical director of Mendelics Analyze Genomics. All other authors declare no conflicts of interest.
Figures
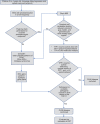
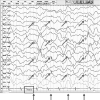
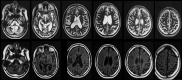
References
-
- Zeman W, Dyken P. Neuronal ceroid-lipofuscinosis (Batten's disease): relationship to amaurotic family idiocy? Pediatrics. 1969;44(04):570–583. - PubMed
-
- Williams R E, Mole S E. New nomenclature and classification scheme for the neuronal ceroid lipofuscinoses. Neurology. 2012;79(02):183–191. - PubMed
-
- Mole S E, Anderson G, Band H A. Clinical challenges and future therapeutic approaches for neuronal ceroid lipofuscinosis. Lancet Neurol. 2019;18(01):107–116. - PubMed
-
- U.S. Food and Drugs Administration. Original approval letter. Accessed April 11, 2021.https://www.accessdata.fda.gov/drugsatfda_docs/appletter/2017/761052Orig...
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

