Safety and immunogenicity of a ChAdOx1 vaccine against Rift Valley fever in UK adults: an open-label, non-randomised, first-in-human phase 1 clinical trial
- PMID: 37060917
- PMCID: PMC7614834
- DOI: 10.1016/S1473-3099(23)00068-3
Safety and immunogenicity of a ChAdOx1 vaccine against Rift Valley fever in UK adults: an open-label, non-randomised, first-in-human phase 1 clinical trial
Abstract
Background: Rift Valley fever is a viral epidemic illness prevalent in Africa that can be fatal or result in debilitating sequelae in humans. No vaccines are available for human use. We aimed to evaluate the safety and immunogenicity of a non-replicating simian adenovirus-vectored Rift Valley fever (ChAdOx1 RVF) vaccine in humans.
Methods: We conducted a phase 1, first-in-human, open-label, dose-escalation trial in healthy adults aged 18-50 years at the Centre for Clinical Vaccinology and Tropical Medicine, Oxford, UK. Participants were required to have no serious comorbidities or previous history of receiving an adenovirus-based vaccine before enrolment. Participants were non-randomly allocated to receive a single ChAdOx1 RVF dose of either 5 × 109 virus particles (vp), 2·5 × 1010 vp, or 5 × 1010 vp administered intramuscularly into the deltoid of their non-dominant arm; enrolment was sequential and administration was staggered to allow for safety to be assessed before progression to the next dose. Primary outcome measures were assessment of adverse events and secondary outcome measures were Rift Valley fever neutralising antibody titres, Rift Valley fever GnGc-binding antibody titres (ELISA), and cellular response (ELISpot), analysed in all participants who received a vaccine. This trial is registered with ClinicalTrials.gov (NCT04754776).
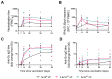
Findings: Between June 11, 2021, and Jan 13, 2022, 15 volunteers received a single dose of either 5 × 109 vp (n=3), 2·5 × 1010 vp (n=6), or 5 × 1010 vp (n=6) ChAdOx1 RVF. Nine participants were female and six were male. 14 (93%) of 15 participants reported solicited local adverse reactions; injection-site pain was the most frequent (13 [87%] of 15). Ten (67%) of 15 participants (from the 2·5 × 1010 vp and 5 × 1010 vp groups only) reported systemic symptoms, which were mostly mild in intensity, the most common being headache (nine [60%] of 15) and fatigue (seven [47%]). All unsolicited adverse events reported within 28 days were either mild or moderate in severity; gastrointestinal symptoms were the most common reaction (at least possibly related to vaccination), occurring in four (27%) of 15 participants. Transient decreases in total white cell, lymphocyte, or neutrophil counts occurred at day 2 in some participants in the intermediate-dose and high-dose groups. Lymphopenia graded as severe occurred in two participants in the 5 × 1010 vp group at a single timepoint, but resolved at the subsequent follow-up visit. No serious adverse events occurred. Rift Valley fever neutralising antibodies were detectable across all dose groups, with all participants in the 5 × 1010 vp dose group having high neutralising antibody titres that peaked at day 28 after vaccination and persisted through the 3-month follow-up. High titres of binding IgG targeting Gc glycoprotein were detected whereas those targeting Gn were comparatively low. IFNγ cellular responses against Rift Valley fever Gn and Gc glycoproteins were observed in all participants except one in the 5 × 1010 vp dose group. These IFNγ responses peaked at 2 weeks after vaccination, were highest in the 5 × 1010 vp dose group, and tended to be more frequent against the Gn glycoprotein.
Interpretation: ChAdOx1 RVF was safe, well tolerated, and immunogenic when administered as a single dose in this study population. The data support further clinical development of ChAdOx1 RVF for human use.
Funding: UK Department of Health and Social Care through the UK Vaccines Network, Oak Foundation, and the Wellcome Trust.
Translation: For the Swahili translation of the abstract see Supplementary Materials section.
Copyright © 2023 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of interests PMF receives funding from the Brazilian Government (Coordenação de Aperfeiçoamento de Pessoal de Nível Superior) for PhD work and consulting fees from Vaccitech, a company developing ChAdOx1 vectored vaccines. KJE is named as a contributor to a patent relating to ChAdOx1 MERS. TAB receives funding from the Medical Research Council UK. SCG is named as an inventor on the patent covering ChAdOx1 use as a vaccine vector and holds stock in Vaccitech. AVSH has received royalties from the COVID-19 vectored ChAdOx1 vaccine to both himself and his institution, and is named as an inventor on the patent covering ChAdOx1 use as a vaccine vector. All other authors declare no competing interests.
Figures
Comment in
-
A novel Rift Valley fever vaccine.Lancet Infect Dis. 2023 Aug;23(8):887-889. doi: 10.1016/S1473-3099(23)00134-2. Epub 2023 Apr 13. Lancet Infect Dis. 2023. PMID: 37060918 No abstract available.
References
-
- Daubney R, Hudson JR, Granham PC. Enzootic Hepatitis or Rift Valley fever: an undescribed virus disease of sheep, cattle and man from East Africa. Journal of Pathology and Bacteriology. 1931;34:545–79.
-
- Baudin M, Jumaa AM, Jomma HJE, et al. Association of Rift Valley fever virus infection with miscarriage in Sudanese women: a cross-sectional study. Lancet Glob Health. 2016;4(11):e864–e71. - PubMed
-
- Dungu B, Lubisi BA, Ikegami T. Rift Valley fever vaccines: current and future needs. Curr Opin Virol. 2018;29:8–15. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

