SCF/c-Kit-activated signaling and angiogenesis require Gαi1 and Gαi3
- PMID: 37063428
- PMCID: PMC10092767
- DOI: 10.7150/ijbs.82855
SCF/c-Kit-activated signaling and angiogenesis require Gαi1 and Gαi3
Abstract
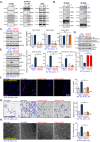
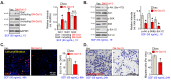
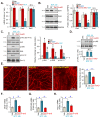
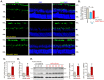
The stem cell factor (SCF) binds to c-Kit in endothelial cells, thus activating downstream signaling and angiogenesis. Herein, we examined the role of G protein subunit alpha inhibitory (Gαi) proteins in this process. In MEFs and HUVECs, Gαi1/3 was associated with SCF-activated c-Kit, promoting c-Kit endocytosis, and binding of key adaptor proteins, subsequently transducing downstream signaling. SCF-induced Akt-mTOR and Erk activation was robustly attenuated by Gαi1/3 silencing or knockout (KO), or due to dominant negative mutations but was strengthened substantially following ectopic overexpression of Gαi1/3. SCF-induced HUVEC proliferation, migration, and capillary tube formation were suppressed after Gαi1/3 silencing or KO, or due to dominant negative mutations. In vivo, endothelial knockdown of Gαi1/3 by intravitreous injection of endothelial-specific shRNA adeno-associated virus (AAV) potently reduced SCF-induced signaling and retinal angiogenesis in mice. Moreover, mRNA and protein expressions of SCF increased significantly in the retinal tissues of streptozotocin-induced diabetic retinopathy (DR) mice. SCF silencing, through intravitreous injection of SCF shRNA AAV, inhibited pathological retinal angiogenesis and degeneration of retinal ganglion cells in DR mice. Finally, the expression of SCF and c-Kit increased in proliferative retinal tissues of human patients with proliferative DR. Taken together, Gαi1/3 mediate SCF/c-Kit-activated signaling and angiogenesis.
© The author(s).
Conflict of interest statement
Competing Interests: The authors have declared that no competing interest exists.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

