A close look at current γδ T-cell immunotherapy
- PMID: 37063836
- PMCID: PMC10102511
- DOI: 10.3389/fimmu.2023.1140623
A close look at current γδ T-cell immunotherapy
Abstract
Owing to their antitumor and major histocompatibility complex (MHC)-independent capacities, γδ T cells have gained popularity in adoptive T-cell immunotherapy in recent years. However, many unknowns still exist regarding γδ T cells, and few clinical data have been collected. Therefore, this review aims to describe all the main features of the applications of γδ T cells and provide a systematic view of current γδ T-cell immunotherapy. Specifically, this review will focus on how γδ T cells performed in treating cancers in clinics, on the γδ T-cell clinical trials that have been conducted to date, and the role of γδ T cells in the pharmaceutical industry.
Keywords: adoptive cell transfer (ACT); cancer immunotherapy; clinical trial; immunotherapy; γδ T cells.
Copyright © 2023 Ma, Feng and Zhou.
Conflict of interest statement
LM, YF, and ZZ are employed by Beijing DCTY Biotech Co., Ltd. LM is also employed as a postdoctoral researcher at Peking University.
Figures

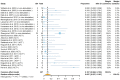
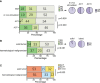
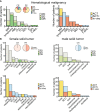

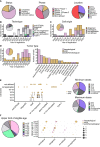
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

