Melatonin suppresses sympathetic vasomotor tone through enhancing GABAA receptor activity in the hypothalamus
- PMID: 37064887
- PMCID: PMC10090494
- DOI: 10.3389/fphys.2023.1166246
Melatonin suppresses sympathetic vasomotor tone through enhancing GABAA receptor activity in the hypothalamus
Abstract
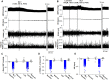
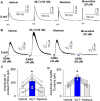
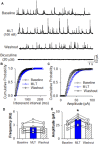
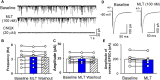
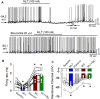
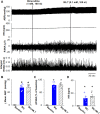
Introduction: Melatonin (5-methoxy-N-acetyl-tryptamine) is a circadian hormone synthesized and secreted by the pineal gland. In addition to regulating circadian rhythms of many physiological functions, melatonin is involved in regulating autonomic nervous function and blood pressure. Hypothalamus paraventricular nucleus (PVN), receiving melatonin projections from the superchiasmatic nucleus, is a critical brain region to regulate neuroendocrine and cardiovascular function. Here, we determined the synaptic mechanisms involved in the effect of melatonin on the sympathetic outflow and blood pressure. Methods and Results: Microinjection of melatonin into the PVN produced a depressor effect and decreased renal sympathetic nerve activity (RSNA). While microinjection of luzindole, a non-selective melatonin receptor antagonist, into the PVN did not change melatonin-induced sympathoinhibition, GABAA receptor antagonist bicuculline eliminated melatonin-induced sympathoinhibition. Furthermore, melatonin decreased firing rate of retrogradely labeled PVN neurons which project to the rostral ventrolateral medulla (RVLM), an effect was not altered by luzindole but eliminated by bicuculline. Melatonin significantly increased the amplitude of spontaneous and evoked GABAergic inhibitory synaptic currents, as well as GABA-induced currents. Conclusion: These data suggest that melatonin in the PVN suppresses sympathetic vasomotor tone through enhancing GABAA receptor activity. This study provides novel information for understanding the cellular mechanisms involved in the effect of melatonin on regulating blood pressure and sympathetic output.
Keywords: GABAA receptor; autonomic nervous system; circadian rhythms; hypothalamus; melatonin; sympathetic nervous system.
Copyright © 2023 Yu, Guo, Jin, Gao, Zheng, Li and Wu.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Pattern of sympathetic vasomotor activity induced by GABAergic inhibition in the brain and spinal cord.Pharmacol Rep. 2020 Feb;72(1):67-79. doi: 10.1007/s43440-019-00025-w. Epub 2020 Jan 8. Pharmacol Rep. 2020. PMID: 32016845
-
Benzodiazepine inhibits hypothalamic presympathetic neurons by potentiation of GABAergic synaptic input.Neuropharmacology. 2007 Feb;52(2):467-75. doi: 10.1016/j.neuropharm.2006.08.024. Epub 2006 Oct 10. Neuropharmacology. 2007. PMID: 17045312
-
Src Kinases Regulate Glutamatergic Input to Hypothalamic Presympathetic Neurons and Sympathetic Outflow in Hypertension.Hypertension. 2017 Jan;69(1):154-162. doi: 10.1161/HYPERTENSIONAHA.116.07947. Epub 2016 Oct 31. Hypertension. 2017. PMID: 27802416 Free PMC article.
-
The paraventricular nucleus: an important component of the central neurocircuitry regulating sympathetic nerve outflow.Acta Physiol Scand. 2003 Jan;177(1):7-15. doi: 10.1046/j.1365-201X.2003.01042.x. Acta Physiol Scand. 2003. PMID: 12492774 Review.
-
Impaired Hypothalamic Regulation of Sympathetic Outflow in Primary Hypertension.Neurosci Bull. 2019 Feb;35(1):124-132. doi: 10.1007/s12264-018-0316-5. Epub 2018 Dec 1. Neurosci Bull. 2019. PMID: 30506315 Free PMC article. Review.
Cited by
-
Neurometabolic alterations in children and adolescents with functional neurological disorder.Neuroimage Clin. 2024;41:103557. doi: 10.1016/j.nicl.2023.103557. Epub 2023 Dec 21. Neuroimage Clin. 2024. PMID: 38219534 Free PMC article.
-
Decreased levels of hydrogen sulfide in the hypothalamic paraventricular nucleus contribute to sympathetic hyperactivity induced by cerebral infarction.Hypertens Res. 2024 May;47(5):1323-1337. doi: 10.1038/s41440-024-01643-5. Epub 2024 Mar 15. Hypertens Res. 2024. PMID: 38491106
-
Melatonin's Impact on Wound Healing.Antioxidants (Basel). 2024 Oct 2;13(10):1197. doi: 10.3390/antiox13101197. Antioxidants (Basel). 2024. PMID: 39456451 Free PMC article. Review.
-
GABA and its receptors' mechanisms in the treatment of insomnia.Heliyon. 2024 Nov 23;10(23):e40665. doi: 10.1016/j.heliyon.2024.e40665. eCollection 2024 Dec 15. Heliyon. 2024. PMID: 39654705 Free PMC article. Review.
-
Dark-phase melatonin administration does not reduce blood pressure but induces changes in parameters related to the control of the cardiovascular system in spontaneously hypertensive rats.Hypertens Res. 2025 Aug;48(8):2218-2233. doi: 10.1038/s41440-025-02247-3. Epub 2025 Jun 9. Hypertens Res. 2025. PMID: 40490489 Free PMC article.
References
LinkOut - more resources
Full Text Sources

