Reprogramming by drug-like molecules leads to regeneration of cochlear hair cell-like cells in adult mice
- PMID: 37068229
- PMCID: PMC10151514
- DOI: 10.1073/pnas.2215253120
Reprogramming by drug-like molecules leads to regeneration of cochlear hair cell-like cells in adult mice
Abstract
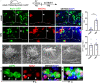
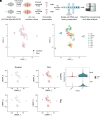
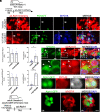
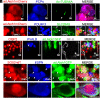
Strategies to overcome irreversible cochlear hair cell (HC) damage and loss in mammals are of vital importance to hearing recovery in patients with permanent hearing loss. In mature mammalian cochlea, co-activation of Myc and Notch1 reprograms supporting cells (SC) and promotes HC regeneration. Understanding of the underlying mechanisms may aid the development of a clinically relevant approach to achieve HC regeneration in the nontransgenic mature cochlea. By single-cell RNAseq, we show that MYC/NICD "rejuvenates" the adult mouse cochlea by activating multiple pathways including Wnt and cyclase activator of cyclic AMP (cAMP), whose blockade suppresses HC-like cell regeneration despite Myc/Notch activation. We screened and identified a combination (the cocktail) of drug-like molecules composing of small molecules and small interfering RNAs to activate the pathways of Myc, Notch1, Wnt and cAMP. We show that the cocktail effectively replaces Myc and Notch1 transgenes and reprograms fully mature wild-type (WT) SCs for HC-like cells regeneration in vitro. Finally, we demonstrate the cocktail is capable of reprogramming adult cochlea for HC-like cells regeneration in WT mice with HC loss in vivo. Our study identifies a strategy by a clinically relevant approach to reprogram mature inner ear for HC-like cells regeneration, laying the foundation for hearing restoration by HC regeneration.
Keywords: RNAseq; adult mouse cochlea; hair cell regeneration; reprogramming; small moleules and siRNA.
Conflict of interest statement
The authors have organizational affiliations to disclose, Z.-Y.C. is a co-founder and a SAB member of Salubritas Therapeutics, which is developing treatments for hearing loss including genome editing, inner ear regeneration, novel delivery, and gene therapy. Z.-Y.C. has ownership of over 5% equity of Salubritas Therapeutics. Y.-Z.Q. and Z.-Y.C. are co-inventors on a patent application that has been filed based on the study by MEE. A patent application on the combination of small molecules/siRNAs in hair cell regeneration has been filed.
Figures



Similar articles
-
Renewed proliferation in adult mouse cochlea and regeneration of hair cells.Nat Commun. 2019 Dec 4;10(1):5530. doi: 10.1038/s41467-019-13157-7. Nat Commun. 2019. PMID: 31797926 Free PMC article.
-
Extensive Supporting Cell Proliferation and Mitotic Hair Cell Generation by In Vivo Genetic Reprogramming in the Neonatal Mouse Cochlea.J Neurosci. 2016 Aug 17;36(33):8734-45. doi: 10.1523/JNEUROSCI.0060-16.2016. J Neurosci. 2016. PMID: 27535918 Free PMC article.
-
Conditional overexpression of neuritin in supporting cells (SCs) mitigates hair cell (HC) damage and induces HC regeneration in the adult mouse cochlea after drug-induced ototoxicity.Hear Res. 2022 Jul;420:108515. doi: 10.1016/j.heares.2022.108515. Epub 2022 May 11. Hear Res. 2022. PMID: 35584572
-
Role of Wnt and Notch signaling in regulating hair cell regeneration in the cochlea.Front Med. 2016 Sep;10(3):237-49. doi: 10.1007/s11684-016-0464-9. Epub 2016 Sep 7. Front Med. 2016. PMID: 27527363 Review.
-
Postnatal development, maturation and aging in the mouse cochlea and their effects on hair cell regeneration.Hear Res. 2013 Mar;297:68-83. doi: 10.1016/j.heares.2012.11.009. Epub 2012 Nov 16. Hear Res. 2013. PMID: 23164734 Free PMC article. Review.
Cited by
-
Thymic Mimetic Cells: Evolutionarily Ancient Mirrors of the Periphery.Immunol Rev. 2025 May;331(1):e70028. doi: 10.1111/imr.70028. Immunol Rev. 2025. PMID: 40247628 Review.
-
Single-cell transcriptional responses of T cells during microsporidia infection.Commun Biol. 2025 Apr 5;8(1):567. doi: 10.1038/s42003-025-07990-4. Commun Biol. 2025. PMID: 40185986 Free PMC article.
-
Potential application value of pigment epithelium-derived factor in sensorineural hearing loss.Front Neurosci. 2023 Dec 14;17:1302124. doi: 10.3389/fnins.2023.1302124. eCollection 2023. Front Neurosci. 2023. PMID: 38164244 Free PMC article. Review.
-
Cell and tissue reprogramming: Unlocking a new era in medical drug discovery.Pharmacol Rev. 2025 Jun 26;77(5):100077. doi: 10.1016/j.pharmr.2025.100077. Online ahead of print. Pharmacol Rev. 2025. PMID: 40683107 Free PMC article. Review.
-
Roles of supporting cells in the maintenance and regeneration of the damaged inner ear: A literature review.J Otol. 2024 Oct;19(4):234-240. doi: 10.1016/j.joto.2024.07.007. Epub 2024 Oct 19. J Otol. 2024. PMID: 39776546 Free PMC article. Review.
References
-
- Corwin J. T., Cotanche D. A., Regeneration of sensory hair cells after acoustic trauma. Science 240, 1772–1774 (1988). - PubMed
-
- Ryals B. M., Rubel E. W., Hair cell regeneration after acoustic trauma in adult Coturnix quail. Science 240, 1774–1776 (1988). - PubMed
-
- Hudspeth A. J., How hearing happens. Neuron 19, 947–950 (1997). - PubMed
-
- Warchol M. E., Lambert P. R., Goldstein B. J., Forge A., Corwin J. T., Regenerative proliferation in inner ear sensory epithelia from adult guinea pigs and humans. Science 259, 1619–1622 (1993). - PubMed
-
- Forge A., Li L., Corwin J. T., Nevill G., Ultrastructural evidence for hair cell regeneration in the mammalian inner ear. Science 259, 1616–1619 (1993). - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

