Integrating Tumor-Intrinsic and Immunologic Factors to Identify Immunogenic Breast Cancers from a Low-Risk Cohort: Results from the Randomized SweBCG91RT Trial
- PMID: 37071498
- PMCID: PMC10150244
- DOI: 10.1158/1078-0432.CCR-22-2746
Integrating Tumor-Intrinsic and Immunologic Factors to Identify Immunogenic Breast Cancers from a Low-Risk Cohort: Results from the Randomized SweBCG91RT Trial
Abstract
Purpose: The local immune infiltrate's influence on tumor progression may be closely linked to tumor-intrinsic factors. The study aimed to investigate whether integrating immunologic and tumor-intrinsic factors can identify patients from a low-risk cohort who may be candidates for radiotherapy (RT) de-escalation.
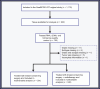
Experimental design: The SweBCG91RT trial included 1,178 patients with stage I to IIA breast cancer, randomized to breast-conserving surgery with or without adjuvant RT, and followed for a median of 15.2 years. We trained two models designed to capture immunologic activity and immunomodulatory tumor-intrinsic qualities, respectively. We then analyzed if combining these two variables could further stratify tumors, allowing for identifying a subgroup where RT de-escalation is feasible, despite clinical indicators of a high risk of ipsilateral breast tumor recurrence (IBTR).
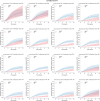
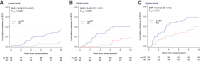
Results: The prognostic effect of the immunologic model could be predicted by the tumor-intrinsic model (Pinteraction = 0.01). By integrating measurements of the immunologic- and tumor-intrinsic models, patients who benefited from an active immune infiltrate could be identified. These patients benefited from standard RT (HR, 0.28; 95% CI, 0.09-0.85; P = 0.025) and had a 5.4% 10-year incidence of IBTR after irradiation despite high-risk genomic indicators and a low frequency of systemic therapy. In contrast, high-risk tumors without an immune infiltrate had a high 10-year incidence of IBTR despite RT treatment (19.5%; 95% CI, 12.2-30.3).
Conclusions: Integrating tumor-intrinsic and immunologic factors may identify immunogenic tumors in early-stage breast cancer populations dominated by ER-positive tumors. Patients who benefit from an activated immune infiltrate may be candidates for RT de-escalation.
©2023 The Authors; Published by the American Association for Cancer Research.
Figures
Comment in
- 1078-0432. doi: 10.1158/1078-0432.CCR-29-9-HI
Similar articles
-
Combining histological grade, TILs, and the PD-1/PD-L1 pathway to identify immunogenic tumors and de-escalate radiotherapy in early breast cancer: a secondary analysis of a randomized clinical trial.J Immunother Cancer. 2023 May;11(5):e006618. doi: 10.1136/jitc-2022-006618. J Immunother Cancer. 2023. PMID: 37208129 Free PMC article. Clinical Trial.
-
Prognostic Factors for Local Control in Breast Cancer After Long-term Follow-up in the EORTC Boost vs No Boost Trial: A Randomized Clinical Trial.JAMA Oncol. 2017 Jan 1;3(1):42-48. doi: 10.1001/jamaoncol.2016.3031. JAMA Oncol. 2017. PMID: 27607734 Clinical Trial.
-
Immune Infiltrate in the Primary Tumor Predicts Effect of Adjuvant Radiotherapy in Breast Cancer; Results from the Randomized SweBCG91RT Trial.Clin Cancer Res. 2021 Feb 1;27(3):749-758. doi: 10.1158/1078-0432.CCR-20-3299. Epub 2020 Nov 4. Clin Cancer Res. 2021. PMID: 33148672 Clinical Trial.
-
Repeat breast-conserving therapy for ipsilateral breast cancer recurrence: A systematic review.Eur J Surg Oncol. 2019 Aug;45(8):1317-1327. doi: 10.1016/j.ejso.2019.02.008. Epub 2019 Feb 10. Eur J Surg Oncol. 2019. PMID: 30795956
-
Local treatment options for young women with ductal carcinoma in situ: A systematic review and meta-analysis comparing breast conserving surgery with or without adjuvant radiotherapy, and mastectomy.Breast. 2022 Jun;63:29-36. doi: 10.1016/j.breast.2022.03.006. Epub 2022 Mar 12. Breast. 2022. PMID: 35299032 Free PMC article.
Cited by
-
Combining histological grade, TILs, and the PD-1/PD-L1 pathway to identify immunogenic tumors and de-escalate radiotherapy in early breast cancer: a secondary analysis of a randomized clinical trial.J Immunother Cancer. 2023 May;11(5):e006618. doi: 10.1136/jitc-2022-006618. J Immunother Cancer. 2023. PMID: 37208129 Free PMC article. Clinical Trial.
-
Identification of high- and low-risk groups for ipsilateral recurrence within 10 years after breast-conserving surgery for ductal carcinoma in situ and personalized treatment: a retrospective study.Gland Surg. 2025 Jul 31;14(7):1213-1229. doi: 10.21037/gs-2025-100. Epub 2025 Jul 28. Gland Surg. 2025. PMID: 40771374 Free PMC article.
-
Predicting immune responsiveness in ER-positive breast cancer for personalized therapy: a population-based study.NPJ Precis Oncol. 2025 Jul 23;9(1):250. doi: 10.1038/s41698-025-01035-z. NPJ Precis Oncol. 2025. PMID: 40702082 Free PMC article.
References
-
- Early Breast Cancer Trialists' Collaborative Group, Darby S, McGale P, Correa C, Taylor C, Arriagada R, et al. . Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet 2011;378:1707–16. - PMC - PubMed
-
- Speers C, Pierce LJ. Postoperative radiotherapy after breast-conserving surgery for early-stage breast cancer: a review. JAMA Oncol 2016;2:1075–82. - PubMed
-
- Schmid P, Cortes J, Pusztai L, McArthur H, Kümmel S, Bergh J, Denkert C, et al. . Pembrolizumab for early triple-negative breast cancer. N Engl J Med 2020;382:810–21. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

