Linearization of the Brevicidine and Laterocidine Lipopeptides Yields Analogues That Retain Full Antibacterial Activity
- PMID: 37071814
- PMCID: PMC10150354
- DOI: 10.1021/acs.jmedchem.3c00308
Linearization of the Brevicidine and Laterocidine Lipopeptides Yields Analogues That Retain Full Antibacterial Activity
Abstract
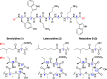
Brevicidine and laterocidine are macrocyclic lipodepsipeptides with selective activity against Gram-negative bacteria, including colistin-resistant strains. Previously, the macrocyclic core of these peptides was thought essential for antibacterial activity. In this study, we show that C-terminal amidation of linear brevicidine and laterocidine scaffolds, and substitution of the native Thr9, yields linear analogues that retain the potent antibacterial activity and low hemolysis of the parent compounds. Furthermore, an alanine scan of both peptides revealed that the aromatic and basic amino acids within the common central scaffold are essential for antibacterial activity. This linearization strategy for modification of cyclic peptides is a highly effective way to reduce the time and cost of peptide synthesis and may be applicable to other non-ribosomal antibacterial peptides.
Conflict of interest statement
The authors declare no competing financial interest.
Figures

References
-
- O’Neill J.Review on Antibacterial Resistance. 2014.
-
- Koo H. B.; Seo J. Antimicrobial peptides under clinical investigation. Pept. Sci. 2019, 111, e24122 10.1002/pep2.24122. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

