Systemic effects of oral tolerance in bone healing
- PMID: 37072616
- PMCID: PMC10113372
- DOI: 10.1038/s41598-023-33591-4
Systemic effects of oral tolerance in bone healing
Abstract
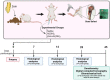
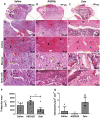
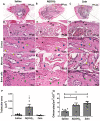
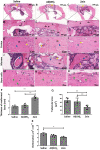
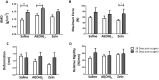
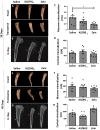
Bone fractures cause acute inflammation that, despite being important for initial repair, may delay the healing of the damaged bone. Parenteral injection of dietary protein has been shown to decrease inflammation and accelerate the repair of skin wounds and other inflammatory pathologies. Thus, our aim was to evaluate whether the intraperitoneal (i.p.) immunization with zein, an abundant protein in rodent chow, would favor bone healing. Wistar rats received i.p. immunization: saline (SG), adjuvant (AG) and zein associated with adjuvant (ZG). Then, a 2 mm of defect bone was performed on the right tibia, and on days 7, 14, 28 and 45 thereafter, analyses were performed. The results showed that the injection of zein reduced inflammation without impairing bone mineralization. Moreover, biomechanical tests demonstrated higher levels of maximum force (N) in ZG, indicating better mechanical resistance in relation to the others. The computerized tomography also indicated lower levels of medullary content in the ZG than in the SG, suggesting the absence of trabeculae in the medullary region in the ZG. These findings suggest that the injection of zein in previously tolerated animals may improve bone repair, leading to mechanically functional bone formation.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Indirect effects of immunological tolerance to a regular dietary protein reduce cutaneous scar formation.Immunology. 2017 Jul;151(3):314-323. doi: 10.1111/imm.12732. Epub 2017 Apr 6. Immunology. 2017. PMID: 28295241 Free PMC article.
-
Subcutaneous injection of an immunologically tolerated protein up to 5 days before skin injuries improves wound healing.Braz J Med Biol Res. 2022 Feb 9;55:e11735. doi: 10.1590/1414-431X2021e11735. eCollection 2022. Braz J Med Biol Res. 2022. PMID: 35170683 Free PMC article.
-
Acceleration of Bone Defect Healing and Regeneration by Low-Intensity Ultrasound Radiation Force in a Rat Tibial Model.Ultrasound Med Biol. 2018 Dec;44(12):2646-2654. doi: 10.1016/j.ultrasmedbio.2018.08.002. Epub 2018 Oct 1. Ultrasound Med Biol. 2018. PMID: 30286949
-
Strontium is incorporated into the fracture callus but does not influence the mechanical strength of healing rat fractures.Calcif Tissue Int. 2011 Feb;88(2):142-52. doi: 10.1007/s00223-010-9439-z. Epub 2010 Dec 9. Calcif Tissue Int. 2011. PMID: 21153023
-
The effects of infrared low-level laser therapy on healing of partial osteotomy of tibia in streptozotocin-induced diabetic rats.Photomed Laser Surg. 2009 Aug;27(4):641-6. doi: 10.1089/pho.2008.2370. Photomed Laser Surg. 2009. PMID: 19694509
Cited by
-
Systemic effects of oral tolerance improve the healing of several and concomitant wounds on different parts of the body.Braz J Med Biol Res. 2025 Apr 14;58:e14689. doi: 10.1590/1414-431X2025e14689. eCollection 2025. Braz J Med Biol Res. 2025. PMID: 40243821 Free PMC article.
-
Non-contact electrical stimulation via a Vector-potential transformer promotes bone healing in drill-hole injury model.J Bone Miner Metab. 2025 Jul;43(4):348-359. doi: 10.1007/s00774-025-01603-0. Epub 2025 Apr 29. J Bone Miner Metab. 2025. PMID: 40301161 Free PMC article.
References
-
- WHO. Life Expectancy at Birth (Years). https://www.who.int/data/gho/data/indicators/indicator-details/GHO/life-.... Accessed 14 Jul 2022.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

