Direct Monitoring of Membrane Fatty Acid Changes and Effects on the Isoleucine/Valine Pathways in an ndgR Deletion Mutant of Streptomyces coelicolor
- PMID: 37072678
- PMCID: PMC10331944
- DOI: 10.4014/jmb.2301.01016
Direct Monitoring of Membrane Fatty Acid Changes and Effects on the Isoleucine/Valine Pathways in an ndgR Deletion Mutant of Streptomyces coelicolor
Abstract
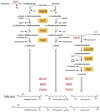
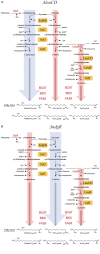
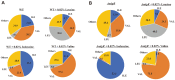
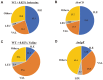
NdgR, a global regulator in soil-dwelling and antibiotic-producing Streptomyces, is known to regulate branched-chain amino acid metabolism by binding to the upstream region of synthetic genes. However, its numerous and complex roles are not yet fully understood. To more fully reveal the function of NdgR, phospholipid fatty acid (PLFA) analysis with gas chromatography-mass spectrometry (GC-MS) was used to assess the effects of an ndgR deletion mutant of Streptomyces coelicolor. The deletion of ndgR was found to decrease the levels of isoleucine- and leucine-related fatty acids but increase those of valine-related fatty acids. Furthermore, the defects in leucine and isoleucine metabolism caused by the deletion impaired the growth of Streptomyces at low temperatures. Supplementation of leucine and isoleucine, however, could complement this defect under cold shock condition. NdgR was thus shown to be involved in the control of branched-chain amino acids and consequently affected the membrane fatty acid composition in Streptomyces. While isoleucine and valine could be synthesized by the same enzymes (IlvB/N, IlvC, IlvD, and IlvE), ndgR deletion did not affect them in the same way. This suggests that NdgR is involved in the upper isoleucine and valine pathways, or that its control over them differs in some respect.
Keywords: Streptomyces coelicolor A3(2); gas chromatography-mass spectrometry; ndgR; phospholipid fatty acid.
Conflict of interest statement
The authors have no financial conflicts of interest to declare.
Figures



Similar articles
-
The role of NdgR in glycerol metabolism in Streptomyces coelicolor.Bioprocess Biosyst Eng. 2017 Oct;40(10):1573-1580. doi: 10.1007/s00449-017-1813-z. Epub 2017 Jul 20. Bioprocess Biosyst Eng. 2017. PMID: 28730324
-
Genome-scale analysis reveals a role for NdgR in the thiol oxidative stress response in Streptomyces coelicolor.BMC Genomics. 2015 Feb 22;16(1):116. doi: 10.1186/s12864-015-1311-0. BMC Genomics. 2015. PMID: 25766138 Free PMC article.
-
NdgR, an IclR-like regulator involved in amino-acid-dependent growth, quorum sensing, and antibiotic production in Streptomyces coelicolor.Appl Microbiol Biotechnol. 2009 Mar;82(3):501-11. doi: 10.1007/s00253-008-1802-x. Epub 2008 Dec 13. Appl Microbiol Biotechnol. 2009. PMID: 19083232
-
Enzymes involved in branched-chain amino acid metabolism in humans.Amino Acids. 2017 Jun;49(6):1005-1028. doi: 10.1007/s00726-017-2412-7. Epub 2017 Mar 21. Amino Acids. 2017. PMID: 28324172 Review.
-
The role of skeletal muscle in the pathogenesis of altered concentrations of branched-chain amino acids (valine, leucine, and isoleucine) in liver cirrhosis, diabetes, and other diseases.Physiol Res. 2021 Jul 12;70(3):293-305. doi: 10.33549/physiolres.934648. Epub 2021 May 12. Physiol Res. 2021. PMID: 33982576 Free PMC article. Review.
Cited by
-
Dual Application of p-Nitrophenol Alkanoate-Based Assay for Soil Selection and Screening of Microbial Strains for Bioplastic Degradation.J Microbiol Biotechnol. 2024 Jul 28;34(7):1530-1543. doi: 10.4014/jmb.2403.03013. Epub 2024 May 30. J Microbiol Biotechnol. 2024. PMID: 38973389 Free PMC article.
-
Moonlighting activity of threonine synthase in cyanobacterial cell death.mSystems. 2025 Jun 17;10(6):e0031025. doi: 10.1128/msystems.00310-25. Epub 2025 May 5. mSystems. 2025. PMID: 40323092 Free PMC article.
References
-
- Bhatia SK, Lee BR, Sathiyanarayanan G, Song HS, Kim J, Jeon JM, et al. Medium engineering for enhanced production of undecylprodigiosin antibiotic in Streptomyces coelicolor using oil palm biomass hydrolysate as a carbon source. Bioresour. Technol. 2016;217:141–149. doi: 10.1016/j.biortech.2016.02.055. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

