Endothelial-to-mesenchymal transition: a precursor to pulmonary arterial remodelling in patients with idiopathic pulmonary fibrosis
- PMID: 37077555
- PMCID: PMC10107070
- DOI: 10.1183/23120541.00487-2022
Endothelial-to-mesenchymal transition: a precursor to pulmonary arterial remodelling in patients with idiopathic pulmonary fibrosis
Abstract
Background: We have previously reported arterial remodelling in patients with idiopathic pulmonary fibrosis (IPF) and suggested that endothelial-to-mesenchymal transition (EndMT) might be central to these changes. This study aims to provide evidence for active EndMT in IPF patients.
Methods: Lung resections from 13 patients with IPF and 15 normal controls (NCs) were immunostained for EndMT biomarkers: vascular endothelial cadherin (VE-cadherin), neural cadherin (N-cadherin), S100A4 and vimentin. Pulmonary arteries were analysed for EndMT markers by using computer- and microscope-assisted image analysis software Image ProPlus7.0. All the analysis was done with observer blinded to subject and diagnosis.
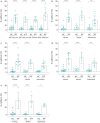
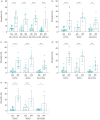
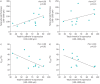
Results: Increased expression of mesenchymal markers N-cadherin (p<0.0001), vimentin (p<0.0001) and S100A4 (p<0.05) was noted with downregulation of junctional endothelial VE-cadherin (p<0.01) in the intimal layer of the arteries from patients with IPF compared to NCs. Cadherin switch was observed in IPF patients, showing increase in endothelial N-cadherin and decrease in VE-cadherin (p<0.01). There was also VE-cadherin shift from junctions to cytoplasm (p<0.01), effecting endothelial cell integrity in patients with IPF. In IPF, individual mesenchymal markers vimentin and N-cadherin negatively correlated with diffusing capacity of the lungs for carbon monoxide (r'= -0.63, p=0.03 and r'= -0.66, p=0.01). Further, N-cadherin positively correlated with arterial thickness (r'=0.58, p=0.03).
Conclusion: This is the first study to demonstrate active EndMT in size-based classified pulmonary arteries from IPF patients and potential role in driving remodelling changes. The mesenchymal markers had a negative impact on the diffusing capacity of the lungs for carbon monoxide. This work also informs early origins of pulmonary hypertension in patients with IPF.
Copyright ©The authors 2023.
Conflict of interest statement
Conflict of interest: S.S. Sohal reports personal fees for lectures from Chiesi, travel support from Chiesi and AstraZeneca, and a research grant from Boehringer Ingelheim outside the submitted work. S.S. Sohal is on the Small Airway Advisory Board for Chiesi Australia. Conflict of interest: All the other authors have no conflict of interest to declare.
Figures





References
LinkOut - more resources
Full Text Sources
Research Materials
