Toward a pan-SARS-CoV-2 vaccine targeting conserved epitopes on spike and non-spike proteins for potent, broad and durable immune responses
- PMID: 37079651
- PMCID: PMC10153712
- DOI: 10.1371/journal.ppat.1010870
Toward a pan-SARS-CoV-2 vaccine targeting conserved epitopes on spike and non-spike proteins for potent, broad and durable immune responses
Abstract
Background: The SARS-CoV-2 non-Spike (S) structural protein targets on nucleocapsid (N), membrane (M) and envelope (E), critical in the host cell interferon response and memory T-cell immunity, are grossly overlooked in COVID vaccine development. The current Spike-only vaccines bear an intrinsic shortfall for promotion of a fuller T cell immunity. Vaccines designed to target conserved epitopes could elicit strong cellular immune responses that would synergize with B cell responses and lead to long-term vaccine success. We pursue a universal (pan-SARS-CoV-2) vaccine against Delta, Omicrons and ever-emergent new mutants.
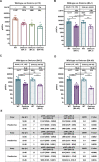
Methods and findings: We explored booster immunogenicity of UB-612, a multitope-vaccine that contains S1-RBD-sFc protein and sequence-conserved promiscuous Th and CTL epitope peptides on the Sarbecovirus N, M and S2 proteins. To a subpopulation (N = 1,478) of infection-free participants (aged 18-85 years) involved in a two-dose Phase-2 trial, a UB-612 booster (third dose) was administered 6-8 months after the second dose. The immunogenicity was evaluated at 14 days post-booster with overall safety monitored until the end of study. The booster induced high viral-neutralizing antibodies against live Wuhan WT (VNT50, 1,711) and Delta (VNT50, 1,282); and against pseudovirus WT (pVNT50, 11,167) vs. Omicron BA.1/BA.2/BA.5 variants (pVNT50, 2,314/1,890/854), respectively. The lower primary neutralizing antibodies in the elderly were uplifted upon boosting to approximately the same high level in young adults. UB-612 also induced potent, durable Th1-oriented (IFN-γ+-) responses (peak/pre-boost/post-boost SFU/106 PBMCs, 374/261/444) along with robust presence of cytotoxic CD8+ T cells (peak/pre-boost/post-boost CD107a+-Granzyme B+, 3.6%/1.8%/1.8%). This UB-612 booster vaccination is safe and well tolerated without SAEs.
Conclusions: By targeting conserved epitopes on viral S2, M and N proteins, UB-612 could provide potent, broad and long-lasting B-cell and T-cell memory immunity and offers the potential as a universal vaccine to fend off Omicrons and new VoCs without resorting to Omicron-specific immunogens.
Trial registration: ClinicalTrials.gov ID: NCT04773067; ClinicalTrials.gov ID: NCT05293665; ClinicalTrials.gov ID: NCT05541861.
Copyright: © 2023 Wang et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
I have read the journal’s policy and the authors of this manuscript have the following competing interests: CYW and WJP are co-inventors on related patent applications. CYW, WJP, BSK, YHH, MSW, YTY, and PYC are employees of UBI Asia.
Figures




Similar articles
-
A multitope SARS-CoV-2 vaccine provides long-lasting B cell and T cell immunity against Delta and Omicron variants.J Clin Invest. 2022 May 16;132(10):e157707. doi: 10.1172/JCI157707. J Clin Invest. 2022. PMID: 35316221 Free PMC article. Clinical Trial.
-
Persistent T cell-mediated immune responses against Omicron variants after the third COVID-19 mRNA vaccine dose.Front Immunol. 2023 Jan 23;14:1099246. doi: 10.3389/fimmu.2023.1099246. eCollection 2023. Front Immunol. 2023. PMID: 36756112 Free PMC article.
-
UB-612 pan-SARS-CoV-2 T cell immunity-promoting vaccine protects against COVID-19 moderate-severe disease.iScience. 2024 Jan 17;27(2):108887. doi: 10.1016/j.isci.2024.108887. eCollection 2024 Feb 16. iScience. 2024. PMID: 38318376 Free PMC article.
-
Identification of B-Cell Epitopes for Eliciting Neutralizing Antibodies against the SARS-CoV-2 Spike Protein through Bioinformatics and Monoclonal Antibody Targeting.Int J Mol Sci. 2022 Apr 14;23(8):4341. doi: 10.3390/ijms23084341. Int J Mol Sci. 2022. PMID: 35457159 Free PMC article. Review.
-
Immune Epitopes of SARS-CoV-2 Spike Protein and Considerations for Universal Vaccine Development.Immunohorizons. 2024 Mar 1;8(3):214-226. doi: 10.4049/immunohorizons.2400003. Immunohorizons. 2024. PMID: 38427047 Free PMC article. Review.
Cited by
-
Beyond the Pandemic Era: Recent Advances and Efficacy of SARS-CoV-2 Vaccines Against Emerging Variants of Concern.Vaccines (Basel). 2025 Apr 17;13(4):424. doi: 10.3390/vaccines13040424. Vaccines (Basel). 2025. PMID: 40333293 Free PMC article. Review.
-
Identification of potential antigenic proteins and epitopes for the development of a monkeypox virus vaccine: an in silico approach.Mol Divers. 2024 Nov 15. doi: 10.1007/s11030-024-11033-1. Online ahead of print. Mol Divers. 2024. PMID: 39546220
-
Molecular basis of potent antiviral HLA-C-restricted CD8+ T cell response to an immunodominant SARS-CoV-2 nucleocapsid epitope.Nat Commun. 2025 Aug 28;16(1):8062. doi: 10.1038/s41467-025-63288-3. Nat Commun. 2025. PMID: 40877317 Free PMC article.
-
Rerouting therapeutic peptides and unlocking their potential against SARS-CoV2.3 Biotech. 2025 May;15(5):116. doi: 10.1007/s13205-025-04270-0. Epub 2025 Apr 4. 3 Biotech. 2025. PMID: 40191455 Review.
-
Safety and immunogenicity of UB-612 heterologous booster in adults primed with mRNA, adenovirus, or inactivated COVID-19 vaccines: a randomized, active-controlled, Phase 3 trial.EClinicalMedicine. 2025 Jul 21;86:103349. doi: 10.1016/j.eclinm.2025.103349. eCollection 2025 Aug. EClinicalMedicine. 2025. PMID: 40727012 Free PMC article.
References
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

