Pyrotinib alone or in combination with docetaxel in refractory HER2-positive gastric cancer: A dose-escalation phase I study
- PMID: 37081722
- PMCID: PMC10225203
- DOI: 10.1002/cam4.5830
Pyrotinib alone or in combination with docetaxel in refractory HER2-positive gastric cancer: A dose-escalation phase I study
Abstract
Aim: Pyrotinib (an irreversible pan-ErbB small-molecular tyrosine kinase inhibitor) was approved in human epidermal growth factor receptor 2 (HER2)-positive breast cancer and showed great antitumor activity in preclinical studies of gastric cancer (GC). This study was first designed to prospectively assess pyrotinib in pretreated HER2-positive GC.
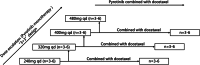
Methods: This multicenter, phase I study followed a standard "3 + 3" design and included two parts. In the pyrotinib part, pyrotinib was administered orally, once per day at dose levels of 240, 320, 400, and 480 mg. In the pyrotinib plus docetaxel part, patients received pyrotinib (qd, d1-21, q3W) combined with docetaxel (60 mg/m2 , d1, q3W) at dose levels of 240, 320, and 400 mg. Primary endpoints were to determine the maximum tolerated dose (MTD) and recommended phase II dose (RP2D) of pyrotinib as monotherapy or coadministered with docetaxel.
Results: A total of 25 patients were enrolled and received pyrotinib (n = 15) or pyrotinib plus docetaxel (n = 10). One DLT was observed in pyrotinib monotherapy part (Grade 3 uncontrolled diarrhea after supportive care) and pyrotinib plus docetaxel part (Grade 4 neutropenia and leukopenia). In the pyrotinib monotherapy part, MTD was not reached. Diarrhea, anemia, neutropenia, and leukopenia were the most common treatment-related adverse events (TRAEs). The RP2D for pyrotinib monotherapy was recommended as 400 mg. After combining with docetaxel, the risk of leukopenia and neutropenia was increased. Grade ≥3 TRAEs were reported for four patients in the monotherapy part and for eight patients in the combination part. Mean t1/2 was approximately 20 h. Pyrotinib exposure was dose-dependent with a nonlinear relationship versus dose. There were five patients who had confirmed partial response (monotherapy: one each at 240, 400, and 480 mg dose cohort; combination therapy: two at 240 mg dose cohort), resulting in an objective response rate of 21% and 20%, respectively.
Conclusions: Pyrotinib alone and combined with docetaxel showed acceptable toxicities in patients with pretreated HER2-positive GC.
Trial registration: This study was registered with ClinicalTrials.gov, NCT02378389.
Keywords: HER2; docetaxel; gastric cancer; phase I study; pyrotinib.
© 2023 The Authors. Cancer Medicine published by John Wiley & Sons Ltd.
Conflict of interest statement
Jianjun Zou and Xiaoyu Zhu are employees of Hengrui. All other authors declare no competing interests.
Figures


Similar articles
-
Neoadjuvant pyrotinib, trastuzumab, and docetaxel for HER2-positive breast cancer (PHEDRA): a double-blind, randomized phase 3 trial.BMC Med. 2022 Dec 27;20(1):498. doi: 10.1186/s12916-022-02708-3. BMC Med. 2022. PMID: 36575513 Free PMC article. Clinical Trial.
-
Phase I Study and Biomarker Analysis of Pyrotinib, a Novel Irreversible Pan-ErbB Receptor Tyrosine Kinase Inhibitor, in Patients With Human Epidermal Growth Factor Receptor 2-Positive Metastatic Breast Cancer.J Clin Oncol. 2017 Sep 20;35(27):3105-3112. doi: 10.1200/JCO.2016.69.6179. Epub 2017 May 12. J Clin Oncol. 2017. PMID: 28498781 Clinical Trial.
-
Pyrotinib versus placebo in combination with trastuzumab and docetaxel as first line treatment in patients with HER2 positive metastatic breast cancer (PHILA): randomised, double blind, multicentre, phase 3 trial.BMJ. 2023 Oct 31;383:e076065. doi: 10.1136/bmj-2023-076065. BMJ. 2023. PMID: 37907210 Free PMC article. Clinical Trial.
-
Neoadjuvant Pyrotinib plus Trastuzumab, Docetaxel, and Carboplatin in Early or Locally Advanced Human Epidermal Receptor 2-Positive Breast Cancer in China: A Multicenter, Randomized, Double-Blind, Placebo-Controlled Phase 2 Trial.Oncol Res Treat. 2023;46(7-8):303-311. doi: 10.1159/000531492. Epub 2023 Jun 9. Oncol Res Treat. 2023. PMID: 37302393 Clinical Trial.
-
Multicenter phase I dose escalation and expansion study of pyrotinib in combination with camrelizumab and chemotherapy as first-line treatment for HER2-positive advanced gastric and gastroesophageal junction adenocarcinoma.EClinicalMedicine. 2023 Nov 20;66:102314. doi: 10.1016/j.eclinm.2023.102314. eCollection 2023 Dec. EClinicalMedicine. 2023. PMID: 38024480 Free PMC article.
Cited by
-
Pyrotinib targeted EGFR/GRP78 mediated cell apoptosis in high EGFR gene copy number gastric cancer.J Exp Clin Cancer Res. 2025 Aug 19;44(1):245. doi: 10.1186/s13046-025-03485-6. J Exp Clin Cancer Res. 2025. PMID: 40830980 Free PMC article.
-
New clinical trial design in precision medicine: discovery, development and direction.Signal Transduct Target Ther. 2024 Mar 4;9(1):57. doi: 10.1038/s41392-024-01760-0. Signal Transduct Target Ther. 2024. PMID: 38438349 Free PMC article. Review.
-
HER2-targeted therapies beyond breast cancer - an update.Nat Rev Clin Oncol. 2024 Sep;21(9):675-700. doi: 10.1038/s41571-024-00924-9. Epub 2024 Jul 22. Nat Rev Clin Oncol. 2024. PMID: 39039196 Review.
-
A stage IV high-grade serous ovarian cancer patient carrying ERBB2 amplification and ERBB2 overexpression benefits from late-line pyrotinib treatment: A case report.Sci Prog. 2025 Jul-Sep;108(3):368504251362310. doi: 10.1177/00368504251362310. Epub 2025 Jul 22. Sci Prog. 2025. PMID: 40696842 Free PMC article.
-
Resistance to Anti-HER2 Therapies in Gastrointestinal Malignancies.Cancers (Basel). 2024 Aug 15;16(16):2854. doi: 10.3390/cancers16162854. Cancers (Basel). 2024. PMID: 39199625 Free PMC article. Review.
References
-
- Do‐Youn O, Bang Y‐J. HER2‐targeted therapies – a role beyond breast cancer. Nat Rev Clin Oncol. 2020;17(1):33‐48. - PubMed
-
- Patel TH, Cecchini M. Targeted therapies in advanced gastric cancer. Curr Treat Options Oncol. 2020;21(9):70. - PubMed
-
- Bang YJ, Cutsem EV, Feyereislova A, et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2‐positive advanced gastric or gastroesophageal junction cancer (ToGA): a phase 3, open‐label, randomised controlled trial. Lancet. 2010;376(9742):687‐697. - PubMed
-
- Siena S, Di Bartolomeo M, Raghav K, et al. Trastuzumab deruxtecan (DS‐8201) in patients with HER2‐expressing metastatic colorectal cancer (DESTINY‐CRC01): a multicentre, open‐label, phase 2 trial. Lancet Oncol. 2021;22(6):779‐789. - PubMed
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

