Enantioselective Metal-Catalyzed Reductive Coupling of Alkynes with Carbonyl Compounds and Imines: Convergent Construction of Allylic Alcohols and Amines
- PMID: 37082110
- PMCID: PMC10112658
- DOI: 10.1021/acscatal.2c02444
Enantioselective Metal-Catalyzed Reductive Coupling of Alkynes with Carbonyl Compounds and Imines: Convergent Construction of Allylic Alcohols and Amines
Abstract
The use of alkynes as vinylmetal pronucleophiles in intermolecular enantioselective metal-catalyzed carbonyl and imine reductive couplings to form allylic alcohols and amines is surveyed. Related hydrogen auto-transfer processes, wherein alcohols or amines serve dually as reductants and carbonyl or imine proelectrophiles, also are cataloged, as are applications in target-oriented synthesis. These processes represent an emerging alternative to the use of stoichiometric vinylmetal reagents or Nozaki-Hiyama-Kishi (NHK) reactions in carbonyl and imine alkenylation.
Keywords: Alkyne; Allylic Alcohol; Allylic Amine; Enantioselective; Hydrogen Transfer; Reductive Coupling.
Figures
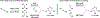


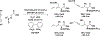

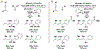

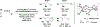

Similar articles
-
Intermolecular Metal-Catalyzed Reductive Coupling of Dienes, Allenes, and Enynes with Carbonyl Compounds and Imines.Chem Rev. 2018 Jun 27;118(12):6026-6052. doi: 10.1021/acs.chemrev.8b00213. Epub 2018 Jun 13. Chem Rev. 2018. PMID: 29897740 Free PMC article. Review.
-
Catalytic Enantioselective Carbonyl Propargylation Beyond Preformed Carbanions: Reductive Coupling and Hydrogen Auto-Transfer.ChemCatChem. 2019 Jan 9;11(1):324-332. doi: 10.1002/cctc.201801121. Epub 2018 Aug 7. ChemCatChem. 2019. PMID: 31588251 Free PMC article.
-
Catalytic Enantioselective Carbonyl Allylation and Propargylation via Alcohol-Mediated Hydrogen Transfer: Merging the Chemistry of Grignard and Sabatier.Acc Chem Res. 2017 Sep 19;50(9):2371-2380. doi: 10.1021/acs.accounts.7b00308. Epub 2017 Aug 9. Acc Chem Res. 2017. PMID: 28792731 Free PMC article.
-
Preparation of stereodefined homoallylic amines from the reductive cross-coupling of allylic alcohols with imines.J Org Chem. 2010 Dec 3;75(23):8048-59. doi: 10.1021/jo101535d. Epub 2010 Nov 11. J Org Chem. 2010. PMID: 21069994 Free PMC article.
-
Historical perspective on ruthenium-catalyzed hydrogen transfer and survey of enantioselective hydrogen auto-transfer processes for the conversion of lower alcohols to higher alcohols.Chem Sci. 2022 Oct 26;13(43):12625-12633. doi: 10.1039/d2sc05621f. eCollection 2022 Nov 9. Chem Sci. 2022. PMID: 36516346 Free PMC article. Review.
Cited by
-
Heteroaryl-Directed Iridium-Catalyzed Enantioselective C-H Alkenylations of Secondary Alcohols.J Am Chem Soc. 2025 Jan 8;147(1):118-124. doi: 10.1021/jacs.4c16414. Epub 2024 Dec 23. J Am Chem Soc. 2025. PMID: 39715233 Free PMC article.
-
Leveraging the Stereochemical Complexity of Octahedral Diastereomeric-at-Metal Catalysts to Unlock Regio-, Diastereo-, and Enantioselectivity in Alcohol-Mediated C-C Couplings via Hydrogen Transfer.J Am Chem Soc. 2024 Mar 27;146(12):7905-7914. doi: 10.1021/jacs.4c01857. Epub 2024 Mar 13. J Am Chem Soc. 2024. PMID: 38478891 Free PMC article. Review.
-
Correction: Synthesis of vinyl-substituted alcohols using acetylene as a C2 building block.Chem Sci. 2023 Feb 6;14(7):1919. doi: 10.1039/d3sc90023a. eCollection 2023 Feb 15. Chem Sci. 2023. PMID: 36812102 Free PMC article.
-
Chiral aldehyde-nickel dual catalysis enables asymmetric α-propargylation of amino acids and stereodivergent synthesis of NP25302.Nat Commun. 2022 Nov 26;13(1):7290. doi: 10.1038/s41467-022-35062-2. Nat Commun. 2022. PMID: 36435942 Free PMC article.
-
Modular α-tertiary amino ester synthesis through cobalt-catalysed asymmetric aza-Barbier reaction.Nat Chem. 2024 Mar;16(3):398-407. doi: 10.1038/s41557-023-01378-9. Epub 2023 Dec 11. Nat Chem. 2024. PMID: 38082178
References
-
- For an introduction to the broad topic of carbonyl and imine addition, see: Comprehensive Organic Synthesis, 2nd ed.; Knochel P; Molander GA, Eds.; Elsevier: Oxford, 2014; Vols. 1 and 2.
-
- For selected reviews on the catalytic enantioselective addition of vinyl anions to carbonyl compounds and imines, see: Wipf P; Kendall C Novel Applications of Alkenyl Zirconocenes. Chem. Eur. J 2002, 8, 1778–1784. - PubMed
- Kauffman MC; Walsh PJ In Science of Synthesis: Stereoselective Synthesis; Molander GA, Ed.; Georg Thieme Verlag KG: Stuttgart, 2011; Vol. 2, pp 449–495.
- Lumbroso A; Cooke ML; Breit B Catalytic Asymmetric Synthesis of Allylic Alcohols and Derivatives and their Applications in Organic Synthesis. Angew. Chem. Int. Ed 2013, 52, 1890–1932. - PubMed
-
- For a recent review on catalytic enantioselective vinylzinc additions to aldehydes, see: Bauer T Enantioselective Dialkylzinc-Mediated Alkynylation, Arylation and Alkenylation of Carbonyl Groups. Coord. Chem. Rev 2015, 299, 83–150.
-
- For selected examples of catalytic enantioselective vinylzinc additions to aldehydes, see: Oppolzer W; Radinov RN Catalytic Asymmetric Synthesis of Secondary (E)-Allyl Alcohols from Acetylenes and Aldehydes via (1-Alkenyl)zinc Intermediates. Helv. Chim. Acta 1992, 75, 170–173.
- Oppolzer W; Radinov RN Synthesis of (R)-(−)-Muscone by an Asymmetrically Catalyzed Macrocyclization of an ω-Alkynal. J. Am. Chem. Soc 1993, 115, 1593–1594.
- Soai K; Takahashi K Asymmetric Alkenylation of Chiral and Prochiral Aldehydes Catalysed by Chiral or Achiral Amino Alcohols: Catalytic Diastereoselective Synthesis of Protected erythro-Sphingosine and Enantioselective Synthesis of Chiral Diallyl Alcohols. J. Chem. Soc., Perkin Trans 1 1994, 1257–1258.
- Oppolzer W; Radinov RN; De Brabander J Total Synthesis of the Macrolide (+)-Aspicilin by an Asymmetrically Catalyzed Macrocyclization of an ω-Alkynal Ester. Tetrahedron Lett 1995, 36, 2607–2610.
- Wipf P; Ribe S Zirconocene-Zinc Transmetalation and in Situ Catalytic Asymmetric Addition to Aldehydes. J. Org. Chem 1998, 63, 6454–6455.
- Oppolzer W; Radinov RN; El-Sayed E Catalytic Asymmetric Synthesis of Macrocyclic (E)-Allylic Alcohols from ω-Alkynals via Intramolecular 1-Alkenylzinc/Aldehyde Additions. J. Org. Chem 2001, 66, 4766–4770. - PubMed
- Dahmen S; Bräse S [2,2]Paracyclophane-Based N,O-Ligands in Alkenylzinc Additions to Aldehydes. Org. Lett 2001, 3, 4119–4122. - PubMed
- Chen YK; Lurain AE; Walsh PJ A General, Highly Enantioselective Method for the Synthesis of D and L α-Amino Acids and Allylic Amines. J. Am. Chem. Soc 2002, 124, 12225–12231. - PubMed
- Ji J-X; Qiu L-Q; Yip CW; Chan ASC A Convenient, One-Step Synthesis of Optically Active Tertiary Aminonaphthol and Its Applications in the Highly Enantioselective Alkenylations of Aldehydes. J. Org. Chem 2003, 68, 1589–1590. - PubMed
- Lurain AE; Walsh PJ A Catalytic Asymmetric Method for the Synthesis of γ-Unsaturated β-Amino Acid Derivatives. J. Am. Chem. Soc 2003, 125, 10677–10683. - PubMed
- Jeon S-J; Chen YK; Walsh PJ Catalytic Asymmetric Synthesis of Hydroxy Enol Ethers: Approach to a Two-Carbon Homologation of Aldehydes. Org. Lett 2005, 7, 1729–1732. - PubMed
- Lauterwasser F; Gall J; Höfener S; Bräse S Second-Generation N,O-[2.2]Paracyclophane Ketimine Ligands for the Alkenylzinc Addition to Aliphatic and Aromatic Aldehydes: Scope and Limitations. Adv. Synth. Catal 2006, 348, 2068–2074.
- Salvi L; Jeon S-J; Fisher EL; Carroll PJ; Walsh PJ Catalytic Asymmetric Generation of (Z)-Disubstituted Allylic Alcohols. J. Am. Chem. Soc 2007, 129, 16119–16125. - PMC - PubMed
- DeBerardinis AM; Turlington M; Pu L Activation of Vinyl Iodides for the Highly Enantioselective Addition to Aldehydes. Angew. Chem. Int. Ed 2011, 50, 2368–2370. - PubMed
-
- For selected reviews on enantioselective Nozaki-Hiyama-Kishi vinylations, see: Hargaden GC; Guiry PJ The Development of the Asymmetric Nozaki–Hiyama–Kishi Reaction. Adv. Synth. Catal 2007, 349, 2407–2424.
- Tian Q; Zhang G Recent Advances in the Asymmetric Nozaki–Hiyama–Kishi Reaction. Synthesis 2016, 48, 4038–4049.
- Gil A; Albericio F; AÁlvarez M Role of the Nozaki–Hiyama–Takai–Kishi Reaction in the Synthesis of Natural Products. Chem. Rev 2017, 117, 8420–8446. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
