The efficacy and safety of nirsevimab for the prevention of RSV infection among infants: A systematic review and meta-analysis
- PMID: 37082704
- PMCID: PMC10110918
- DOI: 10.3389/fped.2023.1132740
The efficacy and safety of nirsevimab for the prevention of RSV infection among infants: A systematic review and meta-analysis
Abstract
Background: Respiratory syncytial virus (RSV) is a leading cause of morbidity and mortality among infants with a global incidence of 9.5% and a mortality rate of 2.2%. The management of RSV infection is mainly supportive and, aside from emerging monoclonal antibodies, there has been no benefit of most preventive measures. Recent evidence suggests the potential of nirsevimab in preventing RSV infection.
Objective: This study aims to determine the efficacy and safety of nirsevimab in preventing RSV infection among infants using a review of relevant clinical trials.
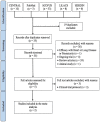
Methods: We performed a random-effects meta-analysis among infants comparing nirsevimab injection vs. placebo. MEDLINE, CENTRAL, Scopus, and ClinicalTrials.gov were searched for relevant trials from inception to June 2022. The selected studies were assessed for risk of bias using the Revised Cochrane Risk-of-Bias (RoB2) tool and for quality of evidence using the Grades of Recommendation, Assessment, Development and Evaluation (GRADE) approach.
Results: Two studies were included. Data analysis showed that among infants, nirsevimab given before the RSV season significantly reduced the risk of medically attended RSV-related infection (RR: 0.26; 95% CI: 0.18-0.38) and the risk of hospitalization due to RSV infection (RR: 0.24; 95% CI: 0.13-0.47). There was no difference in terms of adverse events leading to death (RR: 0.78, 95% CI: 0.20-2.98) and adverse events of special interest (RR: 0.92, 95% CI: 0.25-3.38).
Conclusions: The use of nirsevimab to prevent RSV infections and hospitalization shows its promising potential, but studies on its cost-effectiveness are lacking. We recommend that further studies be done to look into the applicability and cost-effectiveness of nirsevimab.
Keywords: infants; lower respiratory tract infection; nirsevimab; prevention; respiratory syncytial virus.
© 2023 Turalde-Mapili, Mapili, Turalde and Pagcatipunan.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Single-Dose Nirsevimab for Prevention of RSV in Preterm Infants.N Engl J Med. 2020 Jul 30;383(5):415-425. doi: 10.1056/NEJMoa1913556. N Engl J Med. 2020. PMID: 32726528 Clinical Trial.
-
Nirsevimab for Prevention of RSV in Healthy Late-Preterm and Term Infants.N Engl J Med. 2022 Mar 3;386(9):837-846. doi: 10.1056/NEJMoa2110275. N Engl J Med. 2022. PMID: 35235726 Clinical Trial.
-
Early Estimate of Nirsevimab Effectiveness for Prevention of Respiratory Syncytial Virus-Associated Hospitalization Among Infants Entering Their First Respiratory Syncytial Virus Season - New Vaccine Surveillance Network, October 2023-February 2024.MMWR Morb Mortal Wkly Rep. 2024 Mar 7;73(9):209-214. doi: 10.15585/mmwr.mm7309a4. MMWR Morb Mortal Wkly Rep. 2024. PMID: 38457312 Free PMC article.
-
Effectiveness of nirsevimab immunization against RSV infection in preterm infants: a systematic review and meta-analysis.Front Immunol. 2025 Apr 17;16:1581970. doi: 10.3389/fimmu.2025.1581970. eCollection 2025. Front Immunol. 2025. PMID: 40313952 Free PMC article.
-
Breakthrough therapy designation of nirsevimab for the prevention of lower respiratory tract illness caused by respiratory syncytial virus infections (RSV).Expert Opin Investig Drugs. 2022 Jan;31(1):23-29. doi: 10.1080/13543784.2022.2020248. Epub 2021 Dec 28. Expert Opin Investig Drugs. 2022. PMID: 34937485 Review.
Cited by
-
Estimated impact of nirsevimab prophylaxis on the economic burden of respiratory syncytial virus disease in Northern Italy.Ital J Pediatr. 2025 May 21;51(1):151. doi: 10.1186/s13052-025-01991-z. Ital J Pediatr. 2025. PMID: 40400006 Free PMC article.
-
Efficacy and safety of nirsevimab for preventing respiratory syncytial virus infection in infants: an updated systematic review and meta-analysis encompassing 11,001 participants.Proc (Bayl Univ Med Cent). 2025 Apr 16;38(4):524-534. doi: 10.1080/08998280.2025.2488211. eCollection 2025. Proc (Bayl Univ Med Cent). 2025. PMID: 40557197 Free PMC article.
-
Nirsevimab Prophylaxis for Reduction of Respiratory Syncytial Virus Complications in Hospitalised Infants: The Multi-Centre Study During the 2023-2024 Season in Andalusia, Spain (NIRSEGRAND).Vaccines (Basel). 2025 Feb 12;13(2):175. doi: 10.3390/vaccines13020175. Vaccines (Basel). 2025. PMID: 40006722 Free PMC article.
-
Respiratory syncytial virus infections - recent developments providing promising new tools for disease prevention.Euro Surveill. 2023 Dec;28(49):2300686. doi: 10.2807/1560-7917.ES.2023.28.49.2300686. Euro Surveill. 2023. PMID: 38062943 Free PMC article. No abstract available.
-
Systematic Review and Expert Consensus on the Use of Long-acting Monoclonal Antibodies for Prevention of Respiratory Syncytial Virus Disease: ARMADA (Advancing RSV Management And Disease Awareness) Taskforce.Open Forum Infect Dis. 2025 Jul 2;12(7):ofaf396. doi: 10.1093/ofid/ofaf396. eCollection 2025 Jul. Open Forum Infect Dis. 2025. PMID: 40718547 Free PMC article. Review.
References
-
- Li Y, Wang X, Blau DM, Caballero MT, Feikin DR, Gill CJ, et al. Global, regional, and national disease burden estimates of acute lower respiratory infections due to respiratory syncytial virus in children younger than 5 years in 2019: a systematic analysis. Lancet. (2022) 399(10340):2047–64. 10.1016/S0140-6736(22)00478-0 - DOI - PMC - PubMed
-
- Ventre K, Randolph A. Ribavirin for respiratory syncytial virus infection of the lower respiratory tract in infants and young children. In: Ventre K, editors. Cochrane database of systematic reviews. Chichester, UK: John Wiley & Sons, Ltd; (2007):1–19. Available at: https://doi.wiley.com/10.1002/14651858.CD000181.pub3. - DOI - PubMed
-
- Fuller HL, Del Mar C. Immunoglobulin treatment for respiratory syncytial virus infection. In: Fuller HL, editors. Cochrane database of systematic reviews. Chichester, UK: John Wiley & Sons, Ltd; (2006). p. 1–13. Available at: https://doi.wiley.com/10.1002/14651858.CD004883.pub2. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources