The use of commercial fibrin glue in dermal replacement material reduces angiogenic and lymphangiogenic gene and protein expression in vitro
- PMID: 37082911
- PMCID: PMC10226008
- DOI: 10.1177/08853282231171681
The use of commercial fibrin glue in dermal replacement material reduces angiogenic and lymphangiogenic gene and protein expression in vitro
Abstract
Background: Commercial fibrin glue is increasingly finding its way into clinical practice in surgeries to seal anastomosis, and initiate hemostasis or tissue repair. Human biological glue is also being discussed as a possible cell carrier. To date, there are only a few studies addressing the effects of fibrin glue on the cell-molecular level. This study examines the effects of fibrin glue on angiogenesis and lymphangiogenesis, as well as adipose-derived stem cells (ASCs) with a focus on gene and protein expression in scaffolds regularly used for tissue engineering approaches.
Methods: Collagen-based dermal regeneration matrices (DRM) were seeded with human umbilical vein endothelial cells (HUVEC), human dermal lymphatic endothelial cells (LECs), or adipose-derived stem cells (ASC) and fixed with or without fibrin glue according to the experimental group. Cultures were maintained for 1 and 7 days. Finally, angiogenic and lymphangiogenic gene and protein expression were measured with special regard to subtypes of vascular endothelial growth factor (VEGF) and corresponding receptors using Multiplex-qPCR and ELISA assays. In addition, the hypoxia-induced factor 1-alpha (HIF1a) mediated intracellular signaling pathways were included in assessments to analyze a hypoxic encapsulating effect of fibrin polymers.
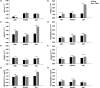
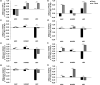
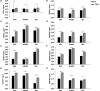
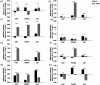
Results: All cell types reacted to fibrin glue application with an alteration of gene and protein expression. In particular, vascular endothelial growth factor A (VEGFA), vascular endothelial growth factor B (VEGFB), vascular endothelial growth factor C (VEGFC), vascular endothelial growth receptor 1 (VEGFR1/FLT1), vascular endothelial growth receptor 2 (VEGFR2/KDR), vascular endothelial growth receptor 3 (VEGFR3/FLT4) and Prospero Homeobox 1 (PROX1) were depressed significantly depending on fibrin glue. Especially short-term fibrin effect led to a continuous downregulation of respective gene and protein expression in HUVECs, LECs, and ASCs.
Conclusion: Our findings demonstrate the impact of fibrin glue application in dermal regeneration with special regard to angiogenesis and lymphangiogenesis. In particular, a short fibrin treatment of 24 hours led to a decrease in gene and protein levels of LECS, HUVECs, and ASCs. In contrast, the long-term application showed less effect on gene and protein expressions. Therefore, this work demonstrated the negative effects of fibrin-treated cells in tissue engineering approaches and could affect wound healing during dermal regeneration.
Keywords: Adipose-derived stem cells; Enzym-linked Immunosorbent Assay; Fibrin glue; Hypoxia-inducible factor 1-alpha; Lymphatic endothelial cells; Multiplex-RT-PCR; Vascular Endothelial Growth Factor; angiogenesis; gene and protein expression; human umbilical vein endothelial cells; lymphangiogenesis.
Conflict of interest statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures


Similar articles
-
Adipose-Derived Stem Cells Improve Angiogenesis and Lymphangiogenesis in a Hypoxic Dermal Regeneration Model In Vitro.Medicina (Kaunas). 2023 Apr 4;59(4):706. doi: 10.3390/medicina59040706. Medicina (Kaunas). 2023. PMID: 37109664 Free PMC article.
-
The mineral dust-induced gene, mdig, regulates angiogenesis and lymphangiogenesis in lung adenocarcinoma by modulating the expression of VEGF-A/C/D via EGFR and HIF-1α signaling.Oncol Rep. 2021 May;45(5):60. doi: 10.3892/or.2021.8011. Epub 2021 Mar 24. Oncol Rep. 2021. PMID: 33760153
-
Use of photosynthetic transgenic cyanobacteria to promote lymphangiogenesis in scaffolds for dermal regeneration.Acta Biomater. 2021 May;126:132-143. doi: 10.1016/j.actbio.2021.03.033. Epub 2021 Mar 20. Acta Biomater. 2021. PMID: 33753313
-
Regulation of VEGFR3 signaling in lymphatic endothelial cells.Front Cell Dev Biol. 2025 Feb 13;13:1527971. doi: 10.3389/fcell.2025.1527971. eCollection 2025. Front Cell Dev Biol. 2025. PMID: 40046235 Free PMC article. Review.
-
Fibrin glue as a drug delivery system.J Control Release. 2010 Nov 20;148(1):49-55. doi: 10.1016/j.jconrel.2010.06.025. Epub 2010 Jul 15. J Control Release. 2010. PMID: 20637815 Free PMC article. Review.
Cited by
-
Bioactivated scaffolds promote angiogenesis and lymphangiogenesis for dermal regeneration in vivo.J Tissue Eng. 2025 Mar 12;16:20417314251317542. doi: 10.1177/20417314251317542. eCollection 2025 Jan-Dec. J Tissue Eng. 2025. PMID: 40078220 Free PMC article.
-
Screening miRNAs to Hinder the Tumorigenesis of Renal Clear Cell Carcinoma Associated with KDR Expression.Curr Cancer Drug Targets. 2025;25(2):183-203. doi: 10.2174/0115680096321287240826065718. Curr Cancer Drug Targets. 2025. PMID: 39289946
References
-
- Canonico S. The use of human fibrin glue in the surgical operations (in eng). Acta Biomed 2003; 74(Suppl 2), pp. 21–25. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

