Targeting neddylation as a novel approach to lung cancer treatment (Review)
- PMID: 37083098
- PMCID: PMC10147314
- DOI: 10.3892/ijo.2023.5513
Targeting neddylation as a novel approach to lung cancer treatment (Review)
Abstract
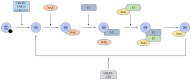
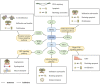
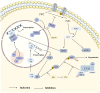
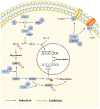
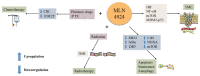
As a protein that resembles ubiquitin, neural precursor cell expressed developmentally downregulated 8 (NEDD8) takes part in neddylation, which modifies substrates in a manner similar to ubiquitination and alters the activity of target proteins. Neddylation may affect the activity of multiple signaling pathways, have a regulatory role in tumor formation, progression and metastasis, and influence the prognosis of cancer treatment. The present review summarizes the regulatory roles of NEDD8 in the MDM2‑p53, NF‑κB, PI3K/AKT/mTOR, hypoxia‑inducible factor, Hippo and receptor tyrosine kinase signaling pathways, as well as in the development and progression of lung cancer.
Keywords: MLN4924; NEDD8; lung cancer; neddylation; signaling pathway; treatment.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
References
-
- Howington JA, Blum MG, Chang AC, Balekian AA, Murthy SC. Treatment of stage I and II non-small cell lung cancer: Diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest. 2013;143(5 Suppl):e278S–e313S. doi: 10.1378/chest.12-2359. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
