Insulin effects on core neurotransmitter pathways involved in schizophrenia neurobiology: a meta-analysis of preclinical studies. Implications for the treatment
- PMID: 37085712
- PMCID: PMC10615753
- DOI: 10.1038/s41380-023-02065-4
Insulin effects on core neurotransmitter pathways involved in schizophrenia neurobiology: a meta-analysis of preclinical studies. Implications for the treatment
Abstract
Impairment of insulin action and metabolic dysregulation have traditionally been associated with schizophrenia, although the molecular basis of such association remains still elusive. The present meta-analysis aims to assess the impact of insulin action manipulations (i.e., hyperinsulinemia, hypoinsulinemia, systemic or brain insulin resistance) on glutamatergic, dopaminergic, γ-aminobutyric acid (GABA)ergic, and serotonergic pathways in the central nervous system. More than one hundred outcomes, including transcript or protein levels, kinetic parameters, and other components of the neurotransmitter pathways, were collected from cultured cells, animals, or humans, and meta-analyzed by applying a random-effects model and adopting Hedges'g to compare means. Two hundred fifteen studies met the inclusion criteria, of which 180 entered the quantitative synthesis. Significant impairments in key regulators of synaptic plasticity processes were detected as the result of insulin handlings. Specifically, protein levels of N-methyl-D-aspartate receptor (NMDAR) subunits including type 2A (NR2A) (Hedges' g = -0.95, 95%C.I. = -1.50, -0.39; p = 0.001; I2 = 47.46%) and 2B (NR2B) (Hedges'g = -0.69, 95%C.I. = -1.35, -0.02; p = 0.043; I2 = 62.09%), and Postsynaptic density protein 95 (PSD-95) (Hedges'g = -0.91, 95%C.I. = -1.51, -0.32; p = 0.003; I2 = 77.81%) were found reduced in insulin-resistant animal models. Moreover, insulin-resistant animals showed significantly impaired dopamine transporter activity, whereas the dopamine D2 receptor mRNA expression (Hedges'g = 3.259; 95%C.I. = 0.497, 6.020; p = 0.021; I2 = 90.61%) increased under insulin deficiency conditions. Insulin action modulated glutamate and GABA release, as well as several enzymes involved in GABA and serotonin synthesis. These results suggest that brain neurotransmitter systems are susceptible to insulin signaling abnormalities, resembling the discrete psychotic disorders' neurobiology and possibly contributing to the development of neurobiological hallmarks of treatment-resistant schizophrenia.
© 2023. The Author(s).
Conflict of interest statement
The author declares no competing interests. The authors have no financial or non-financial interests to disclose with regard to the content and aims of the study therein presented.
Figures
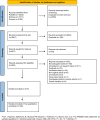


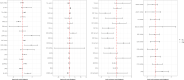


Similar articles
-
Postsynaptic Density-95 Isoform Abnormalities in Schizophrenia.Schizophr Bull. 2017 Jul 1;43(4):891-899. doi: 10.1093/schbul/sbw173. Schizophr Bull. 2017. PMID: 28126896 Free PMC article.
-
Sigma-1 receptor deficiency reduces GABAergic inhibition in the basolateral amygdala leading to LTD impairment and depressive-like behaviors.Neuropharmacology. 2017 Apr;116:387-398. doi: 10.1016/j.neuropharm.2017.01.014. Epub 2017 Jan 17. Neuropharmacology. 2017. PMID: 28108357
-
Mechanism of action of atypical antipsychotic drugs and the neurobiology of schizophrenia.CNS Drugs. 2006;20(5):389-409. doi: 10.2165/00023210-200620050-00004. CNS Drugs. 2006. PMID: 16696579 Review.
-
Regional-specific effects of cerebral ischemia/reperfusion and dehydroepiandrosterone on synaptic NMDAR/PSD-95 complex in male Wistar rats.Brain Res. 2018 Jun 1;1688:73-80. doi: 10.1016/j.brainres.2018.03.023. Epub 2018 Mar 22. Brain Res. 2018. PMID: 29577884
-
Glutamatergic postsynaptic density protein dysfunctions in synaptic plasticity and dendritic spines morphology: relevance to schizophrenia and other behavioral disorders pathophysiology, and implications for novel therapeutic approaches.Mol Neurobiol. 2014 Feb;49(1):484-511. doi: 10.1007/s12035-013-8534-3. Epub 2013 Sep 3. Mol Neurobiol. 2014. PMID: 23999870 Review.
Cited by
-
Codes between Poles: Linking Transcriptomic Insights into the Neurobiology of Bipolar Disorder.Biology (Basel). 2024 Sep 30;13(10):787. doi: 10.3390/biology13100787. Biology (Basel). 2024. PMID: 39452096 Free PMC article.
-
Prevalence and Risk of Schizophrenia and Bipolar Disorder in Patients with Type 1 Diabetes Mellitus: A Systematic Review and Meta-analysis.Indian J Psychol Med. 2025 Mar;47(2):112-118. doi: 10.1177/02537176241238959. Epub 2024 Apr 12. Indian J Psychol Med. 2025. PMID: 39564341 Free PMC article. Review.
-
Addressing brain metabolic connectivity in treatment-resistant schizophrenia: a novel graph theory-driven application of 18F-FDG-PET with antipsychotic dose correction.Schizophrenia (Heidelb). 2024 Dec 19;10(1):116. doi: 10.1038/s41537-024-00535-4. Schizophrenia (Heidelb). 2024. PMID: 39702476 Free PMC article.
-
Relevance of diet in schizophrenia: a review focusing on prenatal nutritional deficiency, obesity, oxidative stress and inflammation.Front Nutr. 2024 Dec 13;11:1497569. doi: 10.3389/fnut.2024.1497569. eCollection 2024. Front Nutr. 2024. PMID: 39734678 Free PMC article.
-
Effects of the selective serotonin reuptake inhibitors citalopram and escitalopram on glucolipid metabolism: a systematic review.Front Endocrinol (Lausanne). 2025 Jun 17;16:1578326. doi: 10.3389/fendo.2025.1578326. eCollection 2025. Front Endocrinol (Lausanne). 2025. PMID: 40600014 Free PMC article.
References
-
- Rummel-Kluge C, Komossa K, Schwarz S, Hunger H, Schmid F, Lobos CA, et al. Head-to-head comparisons of metabolic side effects of second generation antipsychotics in the treatment of schizophrenia: a systematic review and meta-analysis. Schizophr Res. 2010;123:225–33. doi: 10.1016/j.schres.2010.07.012. - DOI - PMC - PubMed
-
- Arranz B, Rosel P, Ramírez N, Dueñas R, Fernández P, Sanchez JM, et al. Insulin resistance and increased leptin concentrations in noncompliant schizophrenia patients but not in antipsychotic-naive first-episode schizophrenia patients. J Clin Psychiatry. 2004;65:1335–42. doi: 10.4088/JCP.v65n1007. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

