Vaccination with recombinant Lactococcus lactis expressing HA1-IgY Fc fusion protein provides protective mucosal immunity against H9N2 avian influenza virus in chickens
- PMID: 37085816
- PMCID: PMC10119832
- DOI: 10.1186/s12985-023-02044-9
Vaccination with recombinant Lactococcus lactis expressing HA1-IgY Fc fusion protein provides protective mucosal immunity against H9N2 avian influenza virus in chickens
Abstract
Background: H9N2 virus is mainly transmitted through the respiratory mucosal pathway, so mucosal immunity is considered to play a good role in controlling avian influenza infection. It is commonly accepted that no adequate mucosal immunity is achieved by inactivated vaccines, which was widely used to prevent and control avian influenza virus infection. Thus, an improved vaccine to induce both mucosal immunity and systemic immunity is urgently required to control H9N2 avian influenza outbreaks in poultry farms.
Methods: In this study, we constructed a novel Lactococcus lactis (L. lactis) strain expressing a recombinant fusion protein consisting of the HA1 proteins derived from an endemic H9N2 virus strain and chicken IgY Fc fragment. We evaluated the immunogenicity and protective efficacy of this recombinant L. lactis HA1-Fc strain.
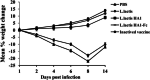
Results: Our data demonstrated that chickens immunized with L. lactis HA1-Fc strain showed significantly increased levels of serum antibodies, mucosal secretory IgA, T cell-mediated immune responses, and lymphocyte proliferation. Furthermore, following challenge with H9N2 avian influenza virus, chickens immunized with L. lactis HA1-Fc strain showed reduced the weight loss, relieved clinical symptoms, and decreased the viral titers and the pathological damage in the lung. Moreover, oropharyngeal and cloacal shedding of the H9N2 influenza virus was detected in chicken immunized with L. lactis HA1-Fc after infection, the results showed the titer was low and reduced quickly to reach undetectable levels at 7 days after infection.
Conclusion: Our data showed that the recombinant L. lactis HA1-Fc strain could induce protective mucosal and systemic immunity, and this study provides a theoretical basis for improving immune responses to prevent and control H9N2 virus infection.
Keywords: H9N2 influenza virus; Immune responses; Lactococcus lactis; Mucosal immune; Oral vaccine.
© 2023. The Author(s).
Conflict of interest statement
The author declares that he has no competing interest.
Figures


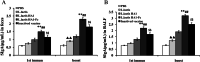
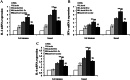



Similar articles
-
Recombinant Lactococcus Lactis Expressing M1-HA2 Fusion Protein Provides Protective Mucosal Immunity Against H9N2 Avian Influenza Virus in Chickens.Front Vet Sci. 2020 Mar 24;7:153. doi: 10.3389/fvets.2020.00153. eCollection 2020. Front Vet Sci. 2020. PMID: 32266297 Free PMC article.
-
Vaccination with recombinant 4 × M2e.HSP70c fusion protein as a universal vaccine candidate enhances both humoral and cell-mediated immune responses and decreases viral shedding against experimental challenge of H9N2 influenza in chickens.Vet Microbiol. 2014 Nov 7;174(1-2):116-26. doi: 10.1016/j.vetmic.2014.09.009. Epub 2014 Sep 23. Vet Microbiol. 2014. PMID: 25293397 Clinical Trial.
-
Vaccine Efficacy of Inactivated, Chimeric Hemagglutinin H9/H5N2 Avian Influenza Virus and Its Suitability for the Marker Vaccine Strategy.J Virol. 2017 Feb 28;91(6):e01693-16. doi: 10.1128/JVI.01693-16. Print 2017 Mar 15. J Virol. 2017. PMID: 28077631 Free PMC article.
-
The immune system of chicken and its response to H9N2 avian influenza virus.Vet Q. 2023 Dec;43(1):1-14. doi: 10.1080/01652176.2023.2228360. Vet Q. 2023. PMID: 37357919 Free PMC article. Review.
-
Lactococcus lactis : A New Strategy for Vaccination.Avicenna J Med Biotechnol. 2017 Oct-Dec;9(4):163-168. Avicenna J Med Biotechnol. 2017. PMID: 29090064 Free PMC article. Review.
Cited by
-
Delivery of dendritic cells targeting 3M2e-HA2 nanoparticles with a CpG adjuvant via lysosomal escape of Salmonella enhances protection against H9N2 avian influenza virus.Poult Sci. 2025 Jan;104(1):104616. doi: 10.1016/j.psj.2024.104616. Epub 2024 Dec 1. Poult Sci. 2025. PMID: 39631272 Free PMC article.
-
Lactobacillus paracasei Expressing Porcine Trefoil Factor 3 and Epidermal Growth Factor: A Novel Approach for Superior Mucosal Repair.Vet Sci. 2025 Apr 14;12(4):365. doi: 10.3390/vetsci12040365. Vet Sci. 2025. PMID: 40284867 Free PMC article.
-
Effect of microencapsulated Fiber2-displaying probiotics loaded with inulin nanoparticles on immunity against fowl adenovirus serotype 4 in chickens.Poult Sci. 2025 Feb;104(2):104762. doi: 10.1016/j.psj.2024.104762. Epub 2025 Jan 1. Poult Sci. 2025. PMID: 39778367 Free PMC article.
-
Surface Display of Avian H5 and H9 Hemagglutinin Antigens on Non-Genetically Modified Lactobacillus Cells for Bivalent Oral AIV Vaccine Development.Microorganisms. 2025 Jul 11;13(7):1649. doi: 10.3390/microorganisms13071649. Microorganisms. 2025. PMID: 40732158 Free PMC article.
-
TonB-Dependent Receptor Protein Displayed on Spores of Bacillus subtilis Stimulates Protective Immune Responses against Acinetobacter baumannii.Vaccines (Basel). 2023 Jun 16;11(6):1106. doi: 10.3390/vaccines11061106. Vaccines (Basel). 2023. PMID: 37376495 Free PMC article.
References
-
- Kishida N, Sakoda Y, Eto M, Sunaga Y, Kida H. Co-infection of Staphylococcus aureus or Haemophilus paragallinarum exacerbates H9N2 influenza A virus infection in chickens. Arch Virol. 2004;11:149. - PubMed
-
- Guo YJ, Krauss S, Senne DA, Mo IP, Lo KS, Xiong XP, et al. Characterization of the pathogenicity of members of the newly established H9N2 influenza virus lineages in Asia. Virology. 2000;2:267. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

