Lumbar endplate microfracture injury induces Modic-like changes, intervertebral disc degeneration and spinal cord sensitization - an in vivo rat model
- PMID: 37086976
- PMCID: PMC10524828
- DOI: 10.1016/j.spinee.2023.04.012
Lumbar endplate microfracture injury induces Modic-like changes, intervertebral disc degeneration and spinal cord sensitization - an in vivo rat model
Abstract
Background context: Endplate (EP) injury plays critical roles in painful IVD degeneration since Modic changes (MCs) are highly associated with pain. Models of EP microfracture that progress to painful conditions are needed to better understand pathophysiological mechanisms and screen therapeutics.
Purpose: Establish in vivo rat lumbar EP microfracture model and assess crosstalk between IVD, vertebra and spinal cord.
Study design/setting: In vivo rat EP microfracture injury model with characterization of IVD degeneration, vertebral remodeling, spinal cord substance P (SubP), and pain-related behaviors.
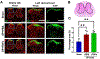
Methods: EP-injury was induced in 5 month-old male Sprague-Dawley rats L4-5 and L5-6 IVDs by puncturing through the cephalad vertebral body and EP into the NP of the IVDs followed by intradiscal injections of TNFα (n=7) or PBS (n=6), compared with Sham (surgery without EP-injury, n=6). The EP-injury model was assessed for IVD height, histological degeneration, pain-like behaviors (hindpaw von Frey and forepaw grip test), lumbar spine MRI and μCT, and spinal cord SubP.
Results: Surgically-induced EP microfracture with PBS and TNFα injection induced IVD degeneration with decreased IVD height and MRI T2 signal, vertebral remodeling, and secondary damage to cartilage EP adjacent to the injury. Both EP injury groups showed MC-like changes around defects with hypointensity on T1-weighted and hyperintensity on T2-weighted MRI, suggestive of MC type 1. EP injuries caused significantly decreased paw withdrawal threshold, reduced axial grip, and increased spinal cord SubP, suggesting axial spinal discomfort and mechanical hypersensitivity and with spinal cord sensitization.
Conclusions: Surgically-induced EP microfracture can cause crosstalk between IVD, vertebra, and spinal cord with chronic pain-like conditions.
Clinical significance: This rat EP microfracture model was validated to induce broad spinal degenerative changes that may be useful to improve understanding of MC-like changes and for therapeutic screening.
Keywords: Endplate microfracture; In vivo rat model; Intervertebral disc degeneration; Modic changes; Pain; Spinal cord sensitization; Spine.
Copyright © 2023 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declarations of Competing Interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures

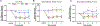




Update of
-
Lumbar endplate microfracture injury induces Modic-like changes, intervertebral disc degeneration and spinal cord sensitization - An In Vivo Rat Model.bioRxiv [Preprint]. 2023 Jan 30:2023.01.27.525924. doi: 10.1101/2023.01.27.525924. bioRxiv. 2023. Update in: Spine J. 2023 Sep;23(9):1375-1388. doi: 10.1016/j.spinee.2023.04.012. PMID: 36778423 Free PMC article. Updated. Preprint.
Similar articles
-
Spinal Cord Sensitization and Spinal Inflammation from an In Vivo Rat Endplate Injury Associated with Painful Intervertebral Disc Degeneration.Int J Mol Sci. 2023 Feb 8;24(4):3425. doi: 10.3390/ijms24043425. Int J Mol Sci. 2023. PMID: 36834838 Free PMC article.
-
Lumbar endplate microfracture injury induces Modic-like changes, intervertebral disc degeneration and spinal cord sensitization - An In Vivo Rat Model.bioRxiv [Preprint]. 2023 Jan 30:2023.01.27.525924. doi: 10.1101/2023.01.27.525924. bioRxiv. 2023. Update in: Spine J. 2023 Sep;23(9):1375-1388. doi: 10.1016/j.spinee.2023.04.012. PMID: 36778423 Free PMC article. Updated. Preprint.
-
Dynamic adaptation of vertebral endplate and trabecular bone following annular injury in a rat model of degenerative disc disease.Spine J. 2018 Nov;18(11):2091-2101. doi: 10.1016/j.spinee.2018.05.045. Epub 2018 Jun 8. Spine J. 2018. PMID: 29886163
-
Mechanisms and clinical implications of intervertebral disc calcification.Nat Rev Rheumatol. 2022 Jun;18(6):352-362. doi: 10.1038/s41584-022-00783-7. Epub 2022 May 9. Nat Rev Rheumatol. 2022. PMID: 35534553 Free PMC article. Review.
-
Association between non-traumatic vertebral fractures and adjacent discs degeneration: a cross-sectional study and literature review.BMC Musculoskelet Disord. 2020 Nov 27;21(1):781. doi: 10.1186/s12891-020-03814-0. BMC Musculoskelet Disord. 2020. PMID: 33246433 Free PMC article. Review.
Cited by
-
Impact of Cage Placement on Vertebral Endplate Cyst Formation and Bone Union in Transforaminal Lumbar Interbody Fusion.Global Spine J. 2025 Apr 28:21925682251339109. doi: 10.1177/21925682251339109. Online ahead of print. Global Spine J. 2025. PMID: 40293858 Free PMC article.
-
Development of Modic Changes After Percutaneous Endoscopic Transforaminal Lumbar Discectomy: From Risk Analysis to Prediction Modeling.J Pain Res. 2024 Dec 17;17:4301-4313. doi: 10.2147/JPR.S475650. eCollection 2024. J Pain Res. 2024. PMID: 39712462 Free PMC article.
-
Animal Models of Disc Degeneration Using Puncture Injury: A 20 Year Perspective.JOR Spine. 2025 Jul 28;8(3):e70093. doi: 10.1002/jsp2.70093. eCollection 2025 Sep. JOR Spine. 2025. PMID: 40727550 Free PMC article. Review.
-
Biomechanical effects of transforaminal endoscopic lumbar discectomy combined with spinal dynamic stabilization system use on adjacent segments: a finite element analysis.BMC Musculoskelet Disord. 2025 Jul 4;26(1):599. doi: 10.1186/s12891-025-08825-3. BMC Musculoskelet Disord. 2025. PMID: 40616028 Free PMC article.
-
Spinal Cord Sensitization and Spinal Inflammation from an In Vivo Rat Endplate Injury Associated with Painful Intervertebral Disc Degeneration.Int J Mol Sci. 2023 Feb 8;24(4):3425. doi: 10.3390/ijms24043425. Int J Mol Sci. 2023. PMID: 36834838 Free PMC article.
References
-
- Battie MC, Videman T, Levalahti E, Gill K, Kaprio J. Genetic and environmental effects on disc degeneration by phenotype and spinal level: a multivariate twin study. Spine (Phila Pa 1976) 2008;33(25):2801–8. - PubMed
-
- Hoy D, March L, Brooks P, et al. The global burden of low back pain: estimates from the Global Burden of Disease 2010 study. Ann Rheum Dis 2014;73(6):968–74. - PubMed
-
- Wang D, Yuan H, Liu A, et al. Analysis of the relationship between the facet fluid sign and lumbar spine motion of degenerative spondylolytic segment using Kinematic MRI. Eur J Radiol 2017;94:6–12. - PubMed
-
- Zehra U, Cheung JPY, Bow C, Lu W, Samartzis D. Multidimensional vertebral endplate defects are associated with disc degeneration, modic changes, facet joint abnormalities, and pain. J Orthop Res 2019;37(5):1080–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

