Lipopolysaccharide structure modulates cationic biocide susceptibility and crystalline biofilm formation in Proteus mirabilis
- PMID: 37089543
- PMCID: PMC10113676
- DOI: 10.3389/fmicb.2023.1150625
Lipopolysaccharide structure modulates cationic biocide susceptibility and crystalline biofilm formation in Proteus mirabilis
Abstract
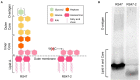
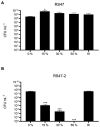
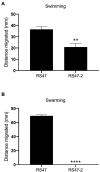
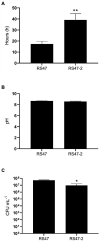
Chlorhexidine (CHD) is a cationic biocide used ubiquitously in healthcare settings. Proteus mirabilis, an important pathogen of the catheterized urinary tract, and isolates of this species are often described as "resistant" to CHD-containing products used for catheter infection control. To identify the mechanisms underlying reduced CHD susceptibility in P. mirabilis, we subjected the CHD tolerant clinical isolate RS47 to random transposon mutagenesis and screened for mutants with reduced CHD minimum inhibitory concentrations (MICs). One mutant recovered from these screens (designated RS47-2) exhibited ~ 8-fold reduction in CHD MIC. Complete genome sequencing of RS47-2 showed a single mini-Tn5 insert in the waaC gene involved in lipopolysaccharide (LPS) inner core biosynthesis. Phenotypic screening of RS47-2 revealed a significant increase in cell surface hydrophobicity and serum susceptibility compared to the wildtype, and confirmed defects in LPS production congruent with waaC inactivation. Disruption of waaC was also associated with increased susceptibility to a range of other cationic biocides but did not affect susceptibility to antibiotics tested. Complementation studies showed that repression of smvA efflux activity in RS47-2 further increased susceptibility to CHD and other cationic biocides, reducing CHD MICs to values comparable with the most CHD susceptible isolates characterized. The formation of crystalline biofilms and blockage of urethral catheters was also significantly attenuated in RS47-2. Taken together, these data show that aspects of LPS structure and upregulation of the smvA efflux system function in synergy to modulate susceptibility to CHD and other cationic biocides, and that LPS structure is also an important factor in P. mirabilis crystalline biofilm formation.
Keywords: AMR; Proteus mirabilis; antimicrobials; biocide; biocide resistance; biofilm; catheter associated urinary tract infection; lipopolysaccharide.
Copyright © 2023 Clarke, Pelling, Bennett, Matsumoto, Gregory, Nzakizwanayo, Slate, Preston, Laabei, Bock, Wand, Ikebukuro, Gebhard, Sutton and Jones.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
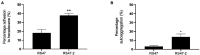
Similar articles
-
Prevalence of mutations associated with tolerance to chlorhexidine and other cationic biocides among Proteus mirabilis clinical isolates.Microbiology (Reading). 2025 Jul;171(7):001580. doi: 10.1099/mic.0.001580. Microbiology (Reading). 2025. PMID: 40694407 Free PMC article.
-
De-repression of the smvA efflux system arises in clinical isolates of Proteus mirabilis and reduces susceptibility to chlorhexidine and other biocides.Antimicrob Agents Chemother. 2019 Sep 9;63(12):e01535-19. doi: 10.1128/AAC.01535-19. Epub 2019 Sep 30. Antimicrob Agents Chemother. 2019. PMID: 31570392 Free PMC article.
-
Identification of mechanisms modulating chlorhexidine and octenidine susceptibility in Proteus mirabilis.J Appl Microbiol. 2024 Jul 2;135(7):lxae173. doi: 10.1093/jambio/lxae173. J Appl Microbiol. 2024. PMID: 38991984
-
Bacterial biofilm formation on indwelling urethral catheters.Lett Appl Microbiol. 2019 Apr;68(4):277-293. doi: 10.1111/lam.13144. Lett Appl Microbiol. 2019. PMID: 30811615 Review.
-
Susceptibility of antibiotic-resistant gram-negative bacteria to biocides: a perspective from the study of catheter biofilms.Symp Ser Soc Appl Microbiol. 2002;(31):163S-170S. Symp Ser Soc Appl Microbiol. 2002. PMID: 12481841 Review.
Cited by
-
Design, synthesis, antimicrobial activity, stability, and mechanism of action of bioresorbable ceragenins.RSC Med Chem. 2025 Jan 21. doi: 10.1039/d4md00990h. Online ahead of print. RSC Med Chem. 2025. PMID: 39897390 Free PMC article.
-
Prevalence of mutations associated with tolerance to chlorhexidine and other cationic biocides among Proteus mirabilis clinical isolates.Microbiology (Reading). 2025 Jul;171(7):001580. doi: 10.1099/mic.0.001580. Microbiology (Reading). 2025. PMID: 40694407 Free PMC article.
-
Unveiling the hidden arsenal: new insights into Proteus mirabilis virulence in UTIs.Front Cell Infect Microbiol. 2024 Nov 13;14:1465460. doi: 10.3389/fcimb.2024.1465460. eCollection 2024. Front Cell Infect Microbiol. 2024. PMID: 39606746 Free PMC article. Review.
-
Use of transcriptomics and genomics to assess the effect of disinfectant exposure on the survival and resistance of Escherichia coli O157:H7, a human pathogen.Front Microbiol. 2024 Oct 23;15:1477683. doi: 10.3389/fmicb.2024.1477683. eCollection 2024. Front Microbiol. 2024. PMID: 39507346 Free PMC article.
References
-
- Alexander Mary K., Miu A., Oh A., Reichelt M., Ho H., Chalouni C., et al. . (2018). Disrupting gram-negative bacterial outer membrane biosynthesis through inhibition of the lipopolysaccharide transporter MsbA. Antimicrob. Agents Chemother. 62, e01142–e01118. doi: 10.1128/AAC.01142-18, PMID: - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

