Prescribing Trends for the Same Patients with Schizophrenia Over 20 Years
- PMID: 37089914
- PMCID: PMC10120815
- DOI: 10.2147/NDT.S390482
Prescribing Trends for the Same Patients with Schizophrenia Over 20 Years
Abstract
Background: Recent pharmacoepidemiology data show an increase in the proportion of patients receiving second-generation antipsychotic (SGA) monotherapy, but no studies have analyzed the same patients over a long period of time. Therefore, in this study, we retrospectively evaluated schizophrenia patients with available data for 20 years to determine whether the drug treatments in the same patients have changed in the past 20 years.
Methods: The study began in April 2021 and was conducted in 15 psychiatric hospitals in Japan. Schizophrenia patients treated in the same hospital for 20 years were retrospectively examined for all prescriptions in 2016, 2011, 2006, and 2001 (ie, every 5 years).
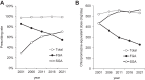
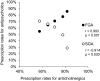
Results: The mean age of the 716 patients surveyed in 2021 was 61.7 years, with 49.0% being female. The rate of antipsychotic monotherapy use showed a slight increasing trend over the past 20 years; the rate of SGA use showed a marked increasing trend from 28.9% to 70.3% over the past 20 years, while the rate of SGA monotherapy use showed a gradual increasing trend over the past 20 years. The rates of concomitant use of anticholinergics, antidepressants, anxiolytics/sleep medications, and mood stabilizers showed decreasing, flat, flat, and flat trends over the past 20 years, respectively.
Conclusion: The results of this study showed a slow but steady substitution of SGAs for first-generation antipsychotics (FGAs) over time, even in the same patients.
Keywords: antipsychotics; pharmacoepidemiology; prescription; schizophrenia; trends.
© 2023 Yasui-Furukori et al.
Conflict of interest statement
Dr. Kensuke Miyazaki reports personal fees from Sumitomo Dainippon Pharma Co., Ltd., outside the submitted work. Professor Kazutaka Shimoda reports grants from Novartis Pharma KK, Dainippon Sumitomo Pharma Co., Astellas Pharma Inc., Meiji Seika Pharma Co., Ltd., Eisai Co., Ltd., Pfizer Inc., Otsuka Pharmaceutical Co., Ltd., Daiichi Sankyo Co., Takeda Pharmaceutical Co., Ltd.; personal fees from Eisai Co., Ltd, Mitsubishi Tanabe Pharma Corporation, Takeda Pharmaceutical Co., Ltd., Meiji Seika Pharma Co., Ltd., Janssen Pharmaceutical Co., Shionogi & Co. Ltd., Dainippon Sumitomo Pharma Co., Daiichi Sankyo Co., and Pfizer Inc., outside the submitted work. The remaining authors report no other conflicts of interest in this work.
Figures
References
-
- NICE. Psychosis and Schizophrenia in Adults: The NICE Guideline on Treatment and Management. NICE Clinical Guideline 178; 2014. Available from: https://www.nice.org.uk/guidance/cg178. Accessed July 29, 2022.
LinkOut - more resources
Full Text Sources
Miscellaneous

