A state-of-the-art review on the MicroRNAs roles in hematopoietic stem cell aging and longevity
- PMID: 37095512
- PMCID: PMC10123996
- DOI: 10.1186/s12964-023-01117-0
A state-of-the-art review on the MicroRNAs roles in hematopoietic stem cell aging and longevity
Abstract
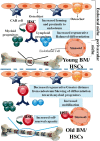
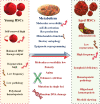
Aging is a biological process determined through time-related cellular and functional impairments, leading to a decreased standard of living for the organism. Recently, there has been an unprecedented advance in the aging investigation, especially the detection that the rate of senescence is at least somewhat regulated via evolutionarily preserved genetic pathways and biological processes. Hematopoietic stem cells (HSCs) maintain blood generation over the whole lifetime of an organism. The senescence process influences many of the natural features of HSC, leading to a decline in their capabilities, independently of their microenvironment. New studies show that HSCs are sensitive to age-dependent stress and gradually lose their self-renewal and regeneration potential with senescence. MicroRNAs (miRNAs) are short, non-coding RNAs that post-transcriptionally inhibit translation or stimulate target mRNA cleavage of target transcripts via the sequence-particular connection. MiRNAs control various biological pathways and processes, such as senescence. Several miRNAs are differentially expressed in senescence, producing concern about their use as moderators of the senescence process. MiRNAs play an important role in the control of HSCs and can also modulate processes associated with tissue senescence in specific cell types. In this review, we display the contribution of age-dependent alterations, including DNA damage, epigenetic landscape, metabolism, and extrinsic factors, which affect HSCs function during aging. In addition, we investigate the particular miRNAs regulating HSCs senescence and age-associated diseases. Video Abstract.
Keywords: Aging; Anti-aging; Hematopoietic stem cell; MicroRNA.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Key miRNAs in Modulating Aging and Longevity: A Focus on Signaling Pathways and Cellular Targets.Curr Mol Pharmacol. 2022;15(5):736-762. doi: 10.2174/1874467214666210917141541. Curr Mol Pharmacol. 2022. PMID: 34533452
-
MicroRNAs as modulators of longevity and the aging process.Hum Genet. 2020 Mar;139(3):291-308. doi: 10.1007/s00439-019-02046-0. Epub 2019 Jul 11. Hum Genet. 2020. PMID: 31297598 Free PMC article. Review.
-
miRNAs in stem cell aging and age-related disease.Mech Ageing Dev. 2017 Dec;168:20-29. doi: 10.1016/j.mad.2017.08.013. Epub 2017 Aug 25. Mech Ageing Dev. 2017. PMID: 28847486 Review.
-
MicroRNAs in hematopoietic stem cell aging.Mech Ageing Dev. 2020 Jul;189:111281. doi: 10.1016/j.mad.2020.111281. Epub 2020 Jun 5. Mech Ageing Dev. 2020. PMID: 32512019 Review.
-
Epigenetic regulation of hematopoietic stem cell aging.Exp Cell Res. 2014 Dec 10;329(2):192-9. doi: 10.1016/j.yexcr.2014.09.013. Epub 2014 Sep 28. Exp Cell Res. 2014. PMID: 25261778 Free PMC article. Review.
Cited by
-
Global microRNA profiling of bone marrow-MSC derived extracellular vesicles identifies miRNAs associated with hematopoietic dysfunction in aplastic anemia.Sci Rep. 2024 Aug 23;14(1):19654. doi: 10.1038/s41598-024-70369-8. Sci Rep. 2024. PMID: 39179703 Free PMC article.
-
Natural Products Treat Colorectal Cancer by Regulating miRNA.Pharmaceuticals (Basel). 2023 Aug 9;16(8):1122. doi: 10.3390/ph16081122. Pharmaceuticals (Basel). 2023. PMID: 37631037 Free PMC article. Review.
-
p63: a crucial player in epithelial stemness regulation.Oncogene. 2023 Nov;42(46):3371-3384. doi: 10.1038/s41388-023-02859-4. Epub 2023 Oct 17. Oncogene. 2023. PMID: 37848625 Free PMC article. Review.
-
miRNA-Mediated Mechanisms in the Generation of Effective and Safe Oncolytic Viruses.Pharmaceutics. 2024 Jul 25;16(8):986. doi: 10.3390/pharmaceutics16080986. Pharmaceutics. 2024. PMID: 39204331 Free PMC article. Review.
-
Epigenetic Mechanisms in Hematologic Aging and Premalignant Conditions.Epigenomes. 2023 Dec 12;7(4):32. doi: 10.3390/epigenomes7040032. Epigenomes. 2023. PMID: 38131904 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

