Enterovirus 71 induces pyroptosis of human neuroblastoma SH-SY5Y cells through miR-146a/ CXCR4 axis
- PMID: 37095967
- PMCID: PMC10121780
- DOI: 10.1016/j.heliyon.2023.e15014
Enterovirus 71 induces pyroptosis of human neuroblastoma SH-SY5Y cells through miR-146a/ CXCR4 axis
Abstract
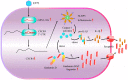
Enterovirus 71 (EV71) is a predominant causative pathogen of hand-foot-and-mouth disease (HFMD) in children. Compared with other HFMD-associated viruses, EV71 tends to induce more severe neurological complications and even death. However, the detailed mechanism of EV71 causes nervous system disorder is still unclear. In this study, we found that EV71 induced the GSDMD/NLRP3-mediated pyroptosis of SH-SY5Y cells through up-regulated miR-146a. Through bioinformatic analysis, we identified C-X-C chemokine receptor type 4 (CXCR4) as the potential target of miR-146a. We noticed that the expression of CXCR4 was regulated by miR-146a during EV71 infection. Moreover, our results show that over-expression of CXCR4 attenuated EV71-induced pyroptosis of SY-SY5Y cells. These results reveal a previously unrecognized mechanism in which EV71 induces nervous system cells damage through regulating miR-146a/CXCR4 mediated pyroptosis.
Keywords: CXCR4; Enterovirus 71 (EV71); Pyroptosis; miR-146a; miRNA.
©2023PublishedbyElsevierLtd.
Conflict of interest statement
The authors declare no competing interests.
Figures




Similar articles
-
Enterovirus 71 Inhibits Pyroptosis through Cleavage of Gasdermin D.J Virol. 2017 Aug 24;91(18):e01069-17. doi: 10.1128/JVI.01069-17. Print 2017 Sep 15. J Virol. 2017. PMID: 28679757 Free PMC article.
-
Transcriptome sequencing analysis of SH-SY5Y cells infected with EV71 reveals the potential neuropathic mechanisms.Virus Res. 2020 Jun;282:197945. doi: 10.1016/j.virusres.2020.197945. Epub 2020 Mar 24. Virus Res. 2020. PMID: 32220619
-
Elevated circulating miR-494 in plasma of children with enterovirus 71 induced hand, foot, and mouth disease and its potential diagnostic value.Acta Virol. 2020;64(3):338-343. doi: 10.4149/av_2020_311. Acta Virol. 2020. PMID: 32985212
-
Enterovirus 71 infection and neurological complications.Korean J Pediatr. 2016 Oct;59(10):395-401. doi: 10.3345/kjp.2016.59.10.395. Epub 2016 Oct 17. Korean J Pediatr. 2016. PMID: 27826325 Free PMC article. Review.
-
Enterovirus 71 infection and vaccines.Clin Exp Vaccine Res. 2017 Jan;6(1):4-14. doi: 10.7774/cevr.2017.6.1.4. Epub 2017 Jan 25. Clin Exp Vaccine Res. 2017. PMID: 28168168 Free PMC article. Review.
Cited by
-
miR-29b-3p regulates cardiomyocytes pyroptosis in CVB3-induced myocarditis through targeting DNMT3A.Cell Mol Biol Lett. 2024 Apr 20;29(1):55. doi: 10.1186/s11658-024-00576-8. Cell Mol Biol Lett. 2024. PMID: 38643118 Free PMC article.
-
CircPTPN11 inhibits the replication of Coxsackievirus B5 through regulating the IFN-I pathway by targeting miR-152-3p/SIRPA axis.Virus Res. 2024 Dec;350:199508. doi: 10.1016/j.virusres.2024.199508. Epub 2024 Dec 12. Virus Res. 2024. PMID: 39647532 Free PMC article.
-
The neuropathological mechanism of EV-A71 infection attributes to inflammatory pryoptosis and viral replication via activating the hsa_circ_0045431/ hsa_miR_584/NLRP3 regulatory axis.Virus Res. 2023 Oct 2;335:199195. doi: 10.1016/j.virusres.2023.199195. Epub 2023 Aug 15. Virus Res. 2023. PMID: 37579846 Free PMC article.
References
LinkOut - more resources
Full Text Sources

