Safety, Immunogenicity, and Efficacy of the NVX-CoV2373 COVID-19 Vaccine in Adolescents: A Randomized Clinical Trial
- PMID: 37099299
- PMCID: PMC10536880
- DOI: 10.1001/jamanetworkopen.2023.9135
Safety, Immunogenicity, and Efficacy of the NVX-CoV2373 COVID-19 Vaccine in Adolescents: A Randomized Clinical Trial
Abstract
Importance: Greater than 20% of cases and 0.4% of deaths from COVID-19 occur in children. Following demonstration of the safety and efficacy of the adjuvanted, recombinant spike protein vaccine NVX-CoV2373 in adults, the PREVENT-19 trial immediately expanded to adolescents.
Objective: To evaluate the safety, immunogenicity, and efficacy of NVX-CoV2373 in adolescents.
Design, setting, and participants: The NVX-CoV2373 vaccine was evaluated in adolescents aged 12 to 17 years in an expansion of PREVENT-19, a phase 3, randomized, observer-blinded, placebo-controlled multicenter clinical trial in the US. Participants were enrolled from April 26 to June 5, 2021, and the study is ongoing. A blinded crossover was implemented after 2 months of safety follow-up to offer active vaccine to all participants. Key exclusion criteria included known previous laboratory-confirmed SARS-CoV-2 infection or known immunosuppression. Of 2304 participants assessed for eligibility, 57 were excluded and 2247 were randomized.
Interventions: Participants were randomized 2:1 to 2 intramuscular injections of NVX-CoV2373 or placebo, 21 days apart.
Main outcomes and measures: Serologic noninferiority of neutralizing antibody responses compared with those in young adults (aged 18-25 years) in PREVENT-19, protective efficacy against laboratory-confirmed COVID-19, and assessment of reactogenicity and safety.
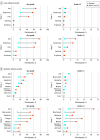
Results: Among 2232 participants (1487 NVX-CoV2373 and 745 placebo recipients), the mean (SD) age was 13.8 (1.4) years, 1172 (52.5%) were male, 1660 (74.4%) were White individuals, and 359 (16.1%) had had a previous SARS-CoV-2 infection at baseline. After vaccination, the ratio of neutralizing antibody geometric mean titers in adolescents compared with those in young adults was 1.5 (95% CI, 1.3-1.7). Twenty mild COVID-19 cases occurred after a median of 64 (IQR, 57-69) days of follow-up, including 6 among NVX-CoV2373 recipients (incidence, 2.90 [95% CI, 1.31-6.46] cases per 100 person-years) and 14 among placebo recipients (incidence, 14.20 [95% CI, 8.42-23.93] cases per 100 person-years), yielding a vaccine efficacy of 79.5% (95% CI, 46.8%-92.1%). Vaccine efficacy for the Delta variant (the only viral variant identified by sequencing [n = 11]) was 82.0% (95% CI, 32.4%-95.2%). Reactogenicity was largely mild to moderate and transient, with a trend toward greater frequency after the second dose of NVX-CoV2373. Serious adverse events were rare and balanced between treatments. No adverse events led to study discontinuation.
Conclusions and relevance: The findings of this randomized clinical trial indicate that NVX-CoV2373 is safe, immunogenic, and efficacious in preventing COVID-19, including the predominant Delta variant, in adolescents.
Trial registration: ClinicalTrials.gov Identifier: NCT04611802.
Conflict of interest statement
Figures


References
-
- US Centers for Disease Control and Prevention . Why children and teens should get vaccinated against COVID-19. Updated December 22, 2022. Accessed November 1, 2022. https://www.cdc.gov/coronavirus/2019-ncov/vaccines/why-vaccinate-childre...
-
- US Centers for Disease Control and Prevention . COVID-19 vaccine recommendations for children and teens. Updated March 2, 2023. Accessed November 1, 2022. https://www.cdc.gov/coronavirus/2019-ncov/vaccines/vaccines-children-tee...
-
- Novavax. First quarter 2022 financial results & operational highlights. May 9, 2022. Accessed November 1, 2022. https://novavax.widen.net/s/zlznqr9b8r/1q22-earnings-slides-final
-
- World Health Organization . COVID-19 vaccines with WHO emergency use listing. Updated May 19, 2022. Accessed November 1, 2022. https://extranet.who.int/pqweb/vaccines/vaccinescovid-19-vaccine-eul-issued
-
- Novavax. Novavax and Serum Institute of India announce first emergency use authorization of Novavax’ COVID-19 vaccine in adolescents ≥12 to <18 in India. March 22, 2022. Accessed November 1, 2022. https://ir.novavax.com/2022-03-22-Novavax-and-Serum-Institute-of-India-A...
Publication types
MeSH terms
Substances
Supplementary concepts
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

