Bioactive lipid regulation of platelet function, hemostasis, and thrombosis
- PMID: 37100208
- PMCID: PMC11143998
- DOI: 10.1016/j.pharmthera.2023.108420
Bioactive lipid regulation of platelet function, hemostasis, and thrombosis
Abstract
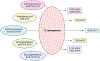
Platelets are small, anucleate cells in the blood that play a crucial role in the hemostatic response but are also implicated in the pathophysiology of cardiovascular disease. It is widely appreciated that polyunsaturated fatty acids (PUFAs) play an integral role in the function and regulation of platelets. PUFAs are substrates for oxygenase enzymes cyclooxygenase-1 (COX-1), 5-lipoxygenase (5-LOX), 12-lipoxygenase (12-LOX) and 15-lipoxygenase (15-LOX). These enzymes generate oxidized lipids (oxylipins) that exhibit either pro- or anti-thrombotic effects. Although the effects of certain oxylipins, such as thromboxanes and prostaglandins, have been studied for decades, only one oxylipin has been therapeutically targeted to treat cardiovascular disease. In addition to the well-known oxylipins, newer oxylipins that demonstrate activity in the platelet have been discovered, further highlighting the expansive list of bioactive lipids that can be used to develop novel therapeutics. This review outlines the known oxylipins, their activity in the platelet, and current therapeutics that target oxylipin signaling.
Keywords: Hemostasis; Oxygenase; Platelet; Prostaglandins; Signal transduction; Thrombosis.
Copyright © 2023 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest Dr. Holinstat is a consultant and equity holder for Veralox Therapeutics and Cereno Scientific. All other authors declare no competing interests for this work.
Figures


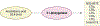

Similar articles
-
The expansive role of oxylipins on platelet biology.J Mol Med (Berl). 2017 Jun;95(6):575-588. doi: 10.1007/s00109-017-1542-4. Epub 2017 May 20. J Mol Med (Berl). 2017. PMID: 28528513 Free PMC article. Review.
-
Regulation of platelet function and thrombosis by omega-3 and omega-6 polyunsaturated fatty acids.Prostaglandins Other Lipid Mediat. 2018 Nov;139:10-18. doi: 10.1016/j.prostaglandins.2018.09.005. Epub 2018 Sep 25. Prostaglandins Other Lipid Mediat. 2018. PMID: 30266534 Free PMC article. Review.
-
12(S)-HETrE, a 12-Lipoxygenase Oxylipin of Dihomo-γ-Linolenic Acid, Inhibits Thrombosis via Gαs Signaling in Platelets.Arterioscler Thromb Vasc Biol. 2016 Oct;36(10):2068-77. doi: 10.1161/ATVBAHA.116.308050. Epub 2016 Jul 28. Arterioscler Thromb Vasc Biol. 2016. PMID: 27470510 Free PMC article.
-
Omega-6 DPA and its 12-lipoxygenase-oxidized lipids regulate platelet reactivity in a nongenomic PPARα-dependent manner.Blood Adv. 2020 Sep 22;4(18):4522-4537. doi: 10.1182/bloodadvances.2020002493. Blood Adv. 2020. PMID: 32946570 Free PMC article.
-
Investigating the catalytic efficiency of C22-Fatty acids with LOX human isozymes and the platelet response of the C22-oxylipin products.Arch Biochem Biophys. 2023 Oct 1;747:109742. doi: 10.1016/j.abb.2023.109742. Epub 2023 Sep 9. Arch Biochem Biophys. 2023. PMID: 37696384 Free PMC article.
Cited by
-
The prognostic and therapeutic significance of polyunsaturated fatty acid-derived oxylipins in ST-segment elevation myocardial infarction.Imeta. 2025 Jan 9;4(1):e266. doi: 10.1002/imt2.266. eCollection 2025 Feb. Imeta. 2025. PMID: 40027487 Free PMC article.
-
Chronic diseases alter the platelet rheostat to promote hyperreactivity and thrombosis.J Clin Invest. 2025 Aug 15;135(16):e194082. doi: 10.1172/JCI194082. eCollection 2025 Aug 15. J Clin Invest. 2025. PMID: 40829178 Free PMC article.
-
The EPA oxylipin, 12-HEPE, directly regulates human platelet activity.J Lipid Res. 2025 May;66(5):100807. doi: 10.1016/j.jlr.2025.100807. Epub 2025 Apr 16. J Lipid Res. 2025. PMID: 40250805 Free PMC article.
-
CYBB identified as a key immune hub gene linking lung cancer and atrial fibrillation.J Cardiothorac Surg. 2025 Jun 18;20(1):262. doi: 10.1186/s13019-025-03510-x. J Cardiothorac Surg. 2025. PMID: 40533809 Free PMC article.
-
Deficiency in platelet 12-lipoxygenase exacerbates inflammation and disease severity during SARS-CoV-2 infection.Proc Natl Acad Sci U S A. 2025 Mar 25;122(12):e2420441122. doi: 10.1073/pnas.2420441122. Epub 2025 Mar 18. Proc Natl Acad Sci U S A. 2025. PMID: 40100623 Free PMC article.
References
-
- Adkins Y, & Kelley DS (2010). Mechanisms underlying the cardioprotective effects of omega-3 polyunsaturated fatty acids. The Journal of Nutritional Biochemistry 21, 781–792. - PubMed
-
- Anand R, & Kaithwas G (2014). Anti-inflammatory potential of alpha-linolenic acid mediated through selective COX inhibition: Computational and experimental data. Inflammation 37, 1297–1306. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

