Efficacy of low-dose rituximab in minimal change disease and prevention of relapse
- PMID: 37101300
- PMCID: PMC10134665
- DOI: 10.1186/s12882-023-03092-7
Efficacy of low-dose rituximab in minimal change disease and prevention of relapse
Abstract
Background: Minimal change disease (MCD) is a major cause of nephrotic syndrome (NS) in children and a minority of adults. The higher tendency to relapse put patients at risk for prolonged exposure to steroids and other immunosuppressive agents. B cell depletion with rituximab (RTX) may be beneficial to the treatment and prevention of frequently relapsing MCD. Therefore, this study aimed to verify the therapeutic/preventive effects of low-dose RTX on the relapse in adult with MCD.
Methods: A total of 33 adult patients were selected for the study, including 22 patients with relapsing MCD in relapse treatment group who were treated with low-dose RTX (200 mg per week × 4 following by 200 mg every 6 months) and 11 patients in relapse prevention group with complete remission (CR) after steroid therapy were treated with RTX (200 mg ×1 every 6 months) for preventing the relapse of MCD.
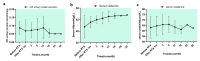
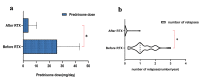
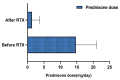
Results: Of the 22 patients with MCD in relapse treatment group, there were 21 cases (95.45%) of remission [2 (9.09%) partial remission (PR), 19 (86.36%) CR], 1 (4.56%) no remission (NR) and 20 (90.90%) relapse-free. The Median duration of sustained remission was 16.3 months (3, 23.5 months, inter quartile range (IQR)). 11 patients in the relapse prevention group during a follow-up of 12 months (9-31 months) had no relapse. The average dose of prednisone in two groups after RTX treatment was significantly lower than before treatment.
Conclusion: The results of this study suggested low-dose RTX can significantly reduce relapse rate and steroid dose in adults with MCD with fewer side effects. Low-dose RTX regimens may be beneficial for the treatment of relapsing MCD in adults and may be the preferred regimen for patients at high risk for the development of adverse events from corticosteroids.
Keywords: Minimal change disease (MCD); Relapse; Rituximab (RTX).
© 2023. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
-
- Avner E, Harmon W, Niaudet P, Yoshikawa N, Emma F, Goldstein S. Idiopathic Nephrotic Syndrome in Children: Clinical Aspects. Pediatric Nephrology, 7th Ed. 2016:pp 2730.
-
- Schnaper H, Robson A, Kopp J. Nephrotic syndrome: Minimal change nephropathy, focal segmental glomerulosclerosis, and collapsing glomerulopathy. In: Diseases of the Kidney and Urinary Tract, 8th ed. 2007:p.1585.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

