Therapeutic Potential of Tumor Metabolic Reprogramming in Triple-Negative Breast Cancer
- PMID: 37108109
- PMCID: PMC10138520
- DOI: 10.3390/ijms24086945
Therapeutic Potential of Tumor Metabolic Reprogramming in Triple-Negative Breast Cancer
Abstract
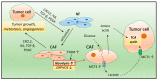
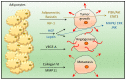
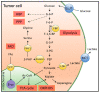
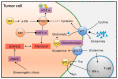
Triple-negative breast cancer (TNBC) is the most aggressive subtype of breast cancer, with clinical features of high metastatic potential, susceptibility to relapse, and poor prognosis. TNBC lacks the expression of the estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2). It is characterized by genomic and transcriptional heterogeneity and a tumor microenvironment (TME) with the presence of high levels of stromal tumor-infiltrating lymphocytes (TILs), immunogenicity, and an important immunosuppressive landscape. Recent evidence suggests that metabolic changes in the TME play a key role in molding tumor development by impacting the stromal and immune cell fractions, TME composition, and activation. Hence, a complex inter-talk between metabolic and TME signaling in TNBC exists, highlighting the possibility of uncovering and investigating novel therapeutic targets. A better understanding of the interaction between the TME and tumor cells, and the underlying molecular mechanisms of cell-cell communication signaling, may uncover additional targets for better therapeutic strategies in TNBC treatment. In this review, we aim to discuss the mechanisms in tumor metabolic reprogramming, linking these changes to potential targetable molecular mechanisms to generate new, physical science-inspired clinical translational insights for the cure of TNBC.
Keywords: cancer metabolism; reprogramming; signaling pathway; therapeutic signaling; triple-negative breast cancer; tumor microenvironment.
Conflict of interest statement
L.S. is an employer at Seagen and holds stocks and shares in Seagen.
Figures
Similar articles
-
Targeting molecular cross-talk between tumor cells and tumor associated macrophage as therapeutic strategy in triple negative breast cancer.Int Immunopharmacol. 2023 Jun;119:110250. doi: 10.1016/j.intimp.2023.110250. Epub 2023 May 8. Int Immunopharmacol. 2023. PMID: 37163922 Review.
-
The role of tumor-infiltrating lymphocytes in triple-negative breast cancer and the research progress of adoptive cell therapy.Front Immunol. 2023 May 18;14:1194020. doi: 10.3389/fimmu.2023.1194020. eCollection 2023. Front Immunol. 2023. PMID: 37275874 Free PMC article. Review.
-
Current Advancements of Plant-Derived Agents for Triple-Negative Breast Cancer Therapy through Deregulating Cancer Cell Functions and Reprogramming Tumor Microenvironment.Int J Mol Sci. 2021 Dec 17;22(24):13571. doi: 10.3390/ijms222413571. Int J Mol Sci. 2021. PMID: 34948368 Free PMC article. Review.
-
Tumor Microenvironment Dynamics of Triple-Negative Breast Cancer Under Radiation Therapy.Int J Mol Sci. 2025 Mar 20;26(6):2795. doi: 10.3390/ijms26062795. Int J Mol Sci. 2025. PMID: 40141437 Free PMC article. Review.
-
Cancer stem cells in TNBC.Semin Cancer Biol. 2022 Jul;82:26-34. doi: 10.1016/j.semcancer.2021.06.015. Epub 2021 Jun 18. Semin Cancer Biol. 2022. PMID: 34147641 Review.
Cited by
-
Identification of intratumoral bacteria that enhance breast tumor metastasis.mBio. 2025 Mar 12;16(3):e0359524. doi: 10.1128/mbio.03595-24. Epub 2025 Feb 11. mBio. 2025. PMID: 39932300 Free PMC article.
-
Triple Negative Breast Cancer: Molecular Subtype-Specific Immune Landscapes with Therapeutic Implications.Cancers (Basel). 2024 May 31;16(11):2094. doi: 10.3390/cancers16112094. Cancers (Basel). 2024. PMID: 38893213 Free PMC article. Review.
-
The Molecular Mechanisms behind Advanced Breast Cancer Metabolism: Warburg Effect, OXPHOS, and Calcium.Front Biosci (Landmark Ed). 2024 Mar 13;29(3):99. doi: 10.31083/j.fbl2903099. Front Biosci (Landmark Ed). 2024. PMID: 38538285 Free PMC article. Review.
-
Repurposing of Strychnine as the Potential Inhibitors of Aldo-keto Reductase Family 1 Members B1 and B10: Computational Modeling and Pharmacokinetic Analysis.Protein J. 2024 Apr;43(2):207-224. doi: 10.1007/s10930-023-10163-z. Epub 2023 Nov 8. Protein J. 2024. PMID: 37940790
-
Nidogen-1 suppresses cell proliferation, migration, and glycolysis via integrin β1-mediated HIF-1α downregulation in triple-negative breast cancer.Sci Rep. 2025 Mar 27;15(1):10633. doi: 10.1038/s41598-024-84880-5. Sci Rep. 2025. PMID: 40148359 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

