Deciphering the Prognostic and Therapeutic Significance of Cell Cycle Regulator CENPF: A Potential Biomarker of Prognosis and Immune Microenvironment for Patients with Liposarcoma
- PMID: 37108172
- PMCID: PMC10139200
- DOI: 10.3390/ijms24087010
Deciphering the Prognostic and Therapeutic Significance of Cell Cycle Regulator CENPF: A Potential Biomarker of Prognosis and Immune Microenvironment for Patients with Liposarcoma
Abstract
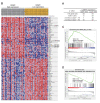
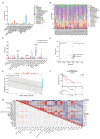
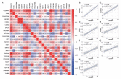
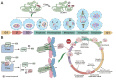
Liposarcoma (LPS) is one of the most common subtypes of sarcoma with a high recurrence rate. CENPF is a regulator of cell cycle, differential expression of which has been shown to be related with various cancers. However, the prognostic value of CENPF in LPS has not been deciphered yet. Using data from TCGA and GEO datasets, the expression difference of CENPF and its effects on the prognosis or immune infiltration of LPS patients were analyzed. As results show, CENPF was significantly upregulated in LPS compared to normal tissues. Survival curves illustrated that high CENPF expression was significantly associated with adverse prognosis. Univariate and multivariate analysis suggested that CENPF expression could be an independent risk factor for LPS. CENPF was closely related to chromosome segregation, microtubule binding and cell cycle. Immune infiltration analysis elucidated a negative correlation between CENPF expression and immune score. In conclusion, CENPF not only could be considered as a potential prognostic biomarker but also a potential malignant indicator of immune infiltration-related survival for LPS. The elevated expression of CENPF reveals an unfavorable prognostic outcome and worse immune score. Thus, therapeutically targeting CENPF combined with immunotherapy might be an attractive strategy for the treatment of LPS.
Keywords: CENPF; cell cycle; immune infiltration; liposarcoma; prognostic biomarker.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Comprehensive Analysis of a Six-Gene Signature Predicting Survival and Immune Infiltration of Liposarcoma Patients and Deciphering Its Therapeutic Significance.Int J Mol Sci. 2024 Jul 16;25(14):7792. doi: 10.3390/ijms25147792. Int J Mol Sci. 2024. PMID: 39063036 Free PMC article.
-
Deciphering the prognostic and therapeutic significance of BAG1 and BAG2 for predicting distinct survival outcome and effects on liposarcoma.Sci Rep. 2024 Oct 4;14(1):23084. doi: 10.1038/s41598-024-67659-6. Sci Rep. 2024. PMID: 39366981 Free PMC article.
-
CENPF Upregulation is Associated with Immunosuppressive Status and Poor Clinical Outcomes in Lung Adenocarcinoma Validated by qRT-PCR.Comb Chem High Throughput Screen. 2024;27(1):78-89. doi: 10.2174/1386207326666230607125353. Comb Chem High Throughput Screen. 2024. PMID: 37287300
-
A novel necroptosis-related gene index for predicting prognosis and a cold tumor immune microenvironment in stomach adenocarcinoma.Front Immunol. 2022 Oct 27;13:968165. doi: 10.3389/fimmu.2022.968165. eCollection 2022. Front Immunol. 2022. PMID: 36389725 Free PMC article. Review.
-
Immunotherapy for liposarcoma: emerging opportunities and challenges.Future Oncol. 2022 Sep;18(30):3449-3461. doi: 10.2217/fon-2021-1549. Epub 2022 Oct 10. Future Oncol. 2022. PMID: 36214331 Review.
Cited by
-
Comprehensive Analysis of a Six-Gene Signature Predicting Survival and Immune Infiltration of Liposarcoma Patients and Deciphering Its Therapeutic Significance.Int J Mol Sci. 2024 Jul 16;25(14):7792. doi: 10.3390/ijms25147792. Int J Mol Sci. 2024. PMID: 39063036 Free PMC article.
-
Centromere Protein F in Tumor Biology: Cancer's Achilles Heel.Cancer Med. 2025 May;14(10):e70949. doi: 10.1002/cam4.70949. Cancer Med. 2025. PMID: 40387105 Free PMC article. Review.
-
Genetic, Epigenetic and Transcriptome Alterations in Liposarcoma for Target Therapy Selection.Cancers (Basel). 2024 Jan 8;16(2):271. doi: 10.3390/cancers16020271. Cancers (Basel). 2024. PMID: 38254762 Free PMC article. Review.
-
Deciphering the prognostic and therapeutic significance of BAG1 and BAG2 for predicting distinct survival outcome and effects on liposarcoma.Sci Rep. 2024 Oct 4;14(1):23084. doi: 10.1038/s41598-024-67659-6. Sci Rep. 2024. PMID: 39366981 Free PMC article.
References
-
- Ng V.Y., Scharschmidt T.J., Mayerson J.L., Fisher J.L. Incidence and survival in sarcoma in the United States: A focus on musculoskeletal lesions. Anticancer Res. 2013;33:2597–2604. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

